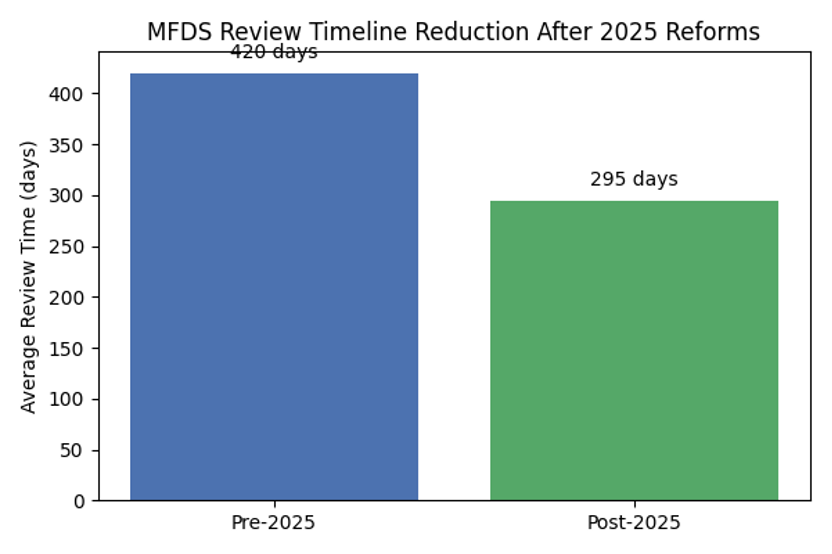
The Korea clinical trial environment is no longer a peripheral consideration. In early 2025 the Korean Ministry of Food and Drug Safety (MFDS) implemented a sweeping overhaul of its new‑drug review system—introducing dedicated review teams, expanding the number of sponsor consultations, allowing rolling review of CTD modules and conducting Good Manufacturing Practice (GMP) evaluations in parallel. These changes aim to cut the average approval timeline from about 420 days to 295 days, signalling that Korea is positioning itself as a strategic hub rather than “one more Asian country” in a regional study.
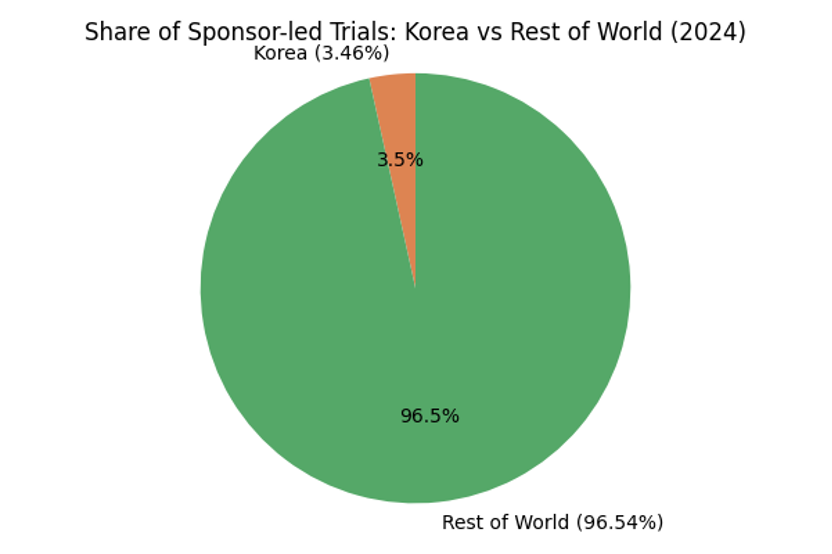
Korea’s ascent is supported by the country’s full listing under the WHO Listed Authority (WLA) framework and longstanding membership in the International Council for Harmonisation (ICH). As a result, Korean sponsor‑led trials already account for roughly 3.46 % of the global total, and data generated in Korea are officially accepted by the U.S. FDA and European EMA because trials adhere to ICH‑GCP standards. Seoul has consistently ranked among the world’s top cities for clinical research, and Korean datasets are routinely used to support submissions in multiple markets. In other words, a well‑designed Korean study can be leveraged for FDA, EMA and MFDS approvals without redundant trials.
Major features of the MFDS 2025 reforms
The table below summarises how the 2025 reforms restructure Korea’s new‑drug review process. Sponsors should note that MFDS fees have increased to fund specialised reviewers and that a dedicated team will stay with a product from the first consultation through final approval.
| Feature | Before 2025 | After 2025 Reforms |
| Review structure | A single lead reviewer dominated the process; cross‑functional collaboration was limited. | A dedicated product‑specific team of about 10–15 experts covers clinical, non‑clinical, CMC, and regulatory disciplines. |
| Consultations | Sponsors typically had about three official face‑to‑face meetings with MFDS. | Sponsors can have up to ten or more meetings, with real‑time discussion of supplementary data and responses. |
| Submission format | The full CTD (Common Technical Document) was submitted at once, and GMP evaluation was conducted separately. | A rolling review allows early submission of CMC and non‑clinical modules, and GMP inspections are conducted in parallel with the document review. |
| Approval timeline | The average review took roughly 420 days. | The target timeline is about 295 days, thanks to rolling review and parallel GMP evaluation. |
Global acceptance and clinical trial strategic planning
Because MFDS is now a WLA and trials in Korea follow ICH‑GCP, foreign regulators legally recognise Korean data if the study is well designed. U.S. 21 CFR 312.120 and EU guidelines permit foreign trials to support approvals when they meet GCP and data integrity requirements. Therefore, sponsors can plan a single Korean trial for multiple markets, aligning endpoints and sample size to satisfy MFDS, FDA and EMA simultaneously. Intoinworld’s article on clinical research management outlines a five‑pillar strategy to make Korean evidence globally submission‑ready; the key elements—data governance, eCTD mapping, label concordance, inspection & quality bridge, and transfer‑package composition—are summarised below.
| Pillar | Core Focus (summary) |
| Data governance | Clearly define data elements and maintain version‑controlled audit trails to ensure traceability and reproducibility. |
| eCTD mapping | Align Korean dossiers with U.S./EU eCTD (electronic Common Technical Document) requirements and perform pre‑validation checks. |
| Label concordance | Synchronize drug labels and diagnostic Instructions‑for‑Use across jurisdictions via structured change‑control procedures. |
| Inspection & quality bridge | Translate Korean inspection findings and corrective actions into the terminology used by foreign regulators, embedding risk‑based monitoring evidence. |
| Transfer package composition | Prepare raw datasets, analysis code, tables/figures, label concordance maps, and change logs so that foreign reviewers can easily reproduce the results. |
Visualizing the reforms
The following charts illustrate the impact of the 2025 reforms and Korea’s growing contribution to global research. The first chart compares the old and new MFDS review timelines; the second shows the share of sponsor‑led trials conducted in Korea vs. the rest of the world in 2024. These visuals reinforce why sponsors should consider Korea a core region in their development strategy.
Why work with a Korean clinical research organization?
Navigating the new MFDS system requires expertise in local regulations, language and culture. While overseas sponsors are not legally obligated to engage a domestic partner, the complexity of 2025 reforms makes local collaboration highly advantageous. A Korea‑based clinical research organization (CRO) such as Intoinworld can tailor protocol design to MFDS expectations, manage consultations and maintain inspection‑ready documentation across sites and data systems. Intoinworld’s guidance on the 2025 review overhaul and its WLA‑era clinical management framework provide deeper insight; visit their article on the MFDS reforms for more details and their piece on clinical research management in the WLA era for best‑practice pillars.
Ready to integrate Korea into your global development plan? Request a quote through Intoinworld’s contact form or explore their newsletter for regular insights on MFDS guidance and clinical research trends. By partnering with a seasoned Korean CRO, you can transform local trials into globally recognized evidence and accelerate access to patients worldwide.





