Current Status of ADC Clinical Trials in Korea
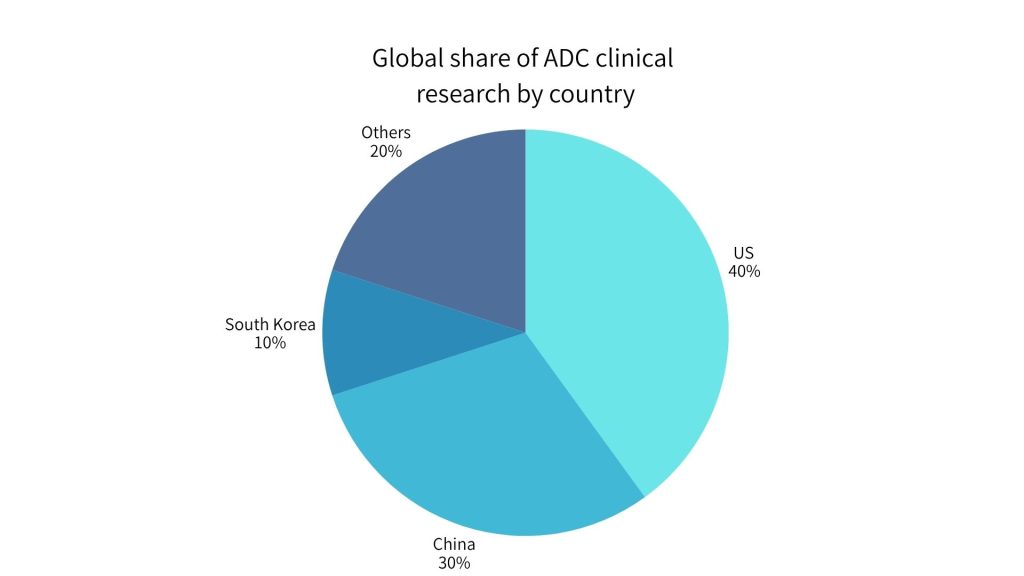
Antibody-drug conjugates (ADCs) are cutting-edge cancer therapies that attach potent chemo-toxins to targeted antibodies, enabling selective destruction of tumor cells while sparing healthy tissue. As global interest in ADCs soars, the market is projected to grow from about $10 billion in 2023 to $28 billion by 2028. South Korea has been quick to ride this wave – the country now accounts for roughly 10% of global ADC clinical research, ranking third worldwide after the United States (40%) and China (30%). Korean pharmaceutical and biotech companies are actively developing ADC pipelines and engaging in co-development partnerships to advance these therapies.
Notably, at the World ADC 2024 conference in San Diego, numerous Korean firms showcased their ADC programs and clinical results to an international audience. Leading the pack, Celltrion forged a partnership with local biotech Pinobio to license an ADC platform and is preparing to file its first ADC investigational new drug (IND) application in 2024.
ABL Bio is developing next-generation bispecific ADCs and gaining global attention for its innovative approach, while companies like Y-Biologics and Intocell are working on B7-H3-targeting ADC candidates in the oncology pipeline. LegoChem Biosciences has demonstrated Korea’s competitive edge by out-licensing its proprietary ADC linker-payload technology to overseas pharma partners, leading to joint clinical trials abroad.
Additionally, major Korean contract manufacturers such as Samsung Biologics and Lotte Biologics are expanding their ADC process development and production capabilities, further strengthening.
ADC Research Trends and Opportunities
ADCs represent a convergence of precise targeting and potent therapy, often hailed as a “magic bullet” in oncology for their ability to deliver chemotherapy directly to cancer cells. By leveraging monoclonal antibodies to hone in on tumor-specific antigens, ADCs can reduce the off-target toxicity common in traditional chemotherapy.
Several ADC therapeutics have already received FDA approval, and their clinical uses are rapidly expanding. South Korean organizations are keeping pace with this global trend, contributing to new ADC clinical data and even achieving success cases of Korea-origin drugs reaching FDA and EMA approvals.
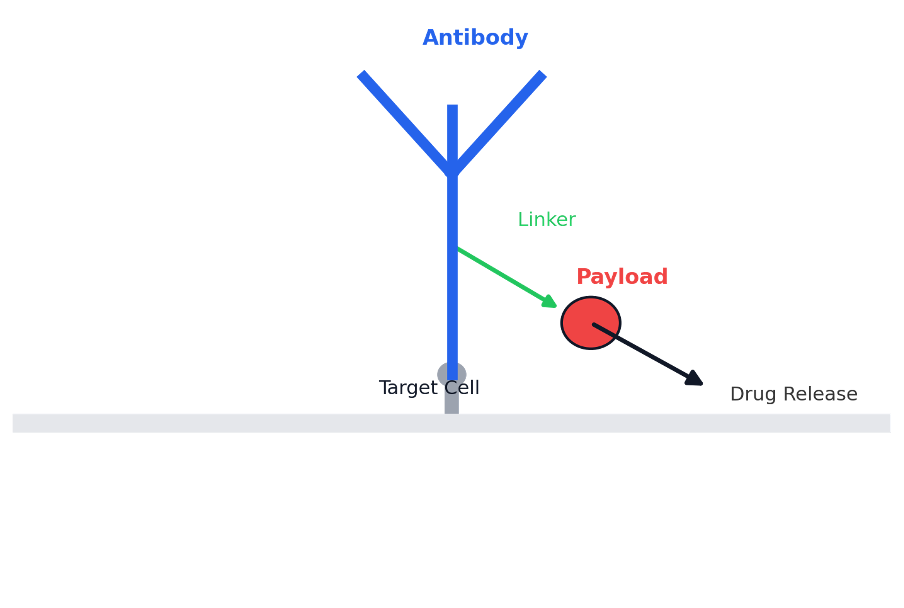
Basic structure of an antibody-drug conjugate (ADC): an antibody (blue) is linked via a chemical linker (green) to a cytotoxic drug payload (red). The antibody targets a tumor cell, delivering the drug payload inside to kill it.
The technology behind ADCs continues to evolve. A prominent area of innovation is bispecific ADCs, which use dual antibodies to target two different antigens simultaneously. This approach aims to overcome tumor resistance mechanisms and enhance efficacy, potentially offering better safety and anti-cancer activity than traditional single-antibody ADCs.
Korean biotech companies like ABL Bio are at the forefront of bispecific ADC research, and others are experimenting with novel linkers and payloads to improve stability and therapeutic windows. Such R&D efforts are expected to diversify the ADC pipeline and create more opportunities for global partnerships and licensing deals in the near future.
Regulatory Innovation and Clinical Infrastructure in Korea
One reason South Korea is emerging as an attractive location for cutting-edge trials like ADCs is its progressive regulatory environment and robust clinical trial infrastructure. In 2025, the Ministry of Food and Drug Safety (MFDS) implemented sweeping reforms to streamline new drug approvals, aiming to shorten the review timeline from the previous average of ~420 days to just 295 days.
Key changes include forming dedicated multi-disciplinary review teams for each drug application, increasing the number of sponsor–regulator meetings (up to 10 meetings for real-time feedback), introducing a rolling review system for continuous data submission, and conducting clinical and GMP inspections in parallel. These innovations have improved the predictability and efficiency of the approval process, helping speed up the development of innovative therapies in Korea.
Korea’s superb medical infrastructure also underpins its rising status. Seoul has been ranked as one of the world’s most active cities for clinical trials, and in 2023 South Korea was the fourth-largest clinical trial hub globally. The country boasts a dense network of top-tier university hospitals and experienced research teams.
Notably, many large hospitals house integrated cancer centers that facilitate multi-disciplinary collaboration, allowing complex trials like those for ADCs to proceed rapidly. Strong government R&D support and advanced IT systems (such as electronic medical records) further enhance patient recruitment and data management. Even during a period when global clinical trial activity declined by 5.5% in 2023, South Korea managed a 9% increase in its number of trials– evidence of the country’s adaptability and innovative drive in clinical research.
Strategic Value of Domestic CROs and CDMOs
Successful drug development requires seamless collaboration between the sponsor (pharmaceutical company), the CRO (contract research organization), and the clinical trial sites (hospitals). A CRO serves as the sponsor’s operational arm in a clinical trial, handling everything from study design and regulatory submissions to site selection, patient monitoring, data management, and biostatistics.
Many resource-limited startups and bioventures rely on CROs to conduct trials that they cannot manage alone, saving time and cost by outsourcing to experts. Globally, CROs have been integral to drug development for over 40 years. In the Korean market, approximately 70% of CRO services are provided by international CRO companies, but the remaining 30% is covered by about 20–30 local CRO firms. This includes Intoinworld, one of the few full-service Korean CROs, which supports both drug and medical device trials with end-to-end project management.
Korean CROs and CDMOs bring strategic advantages to ADC development projects. A local CRO offers on-the-ground expertise tailored to Korea’s regulatory requirements and hospital networks, helping global sponsors navigate approval processes, connect with key investigators, and overcome language or cultural barriers. Intoinworld, for example, provides one-stop clinical trial solutions backed by over a decade of experience – more than half of its projects to date have been in hematology-oncology, the very field where ADCs are prominent.
By partnering with such a domestic CRO, overseas sponsors can initiate Korean trials quickly and efficiently. On the manufacturing side, Korea’s world-class CDMOs like Samsung Biologics and Lotte Biologics complement these efforts by offering high-quality production of biologics, including ADCs for clinical and commercial use. Lotte Biologics has been investing aggressively in ADC manufacturing capacity (establishing a dedicated facility in Syracuse, USA) and collaborating with specialist firms like NJ Bio to build a one-stop ADC CDMO service platform.
This strong CRO–CDMO ecosystem in Korea allows drug developers to integrate their clinical development and manufacturing strategies, paving the way for smoother scale-up from trials to market.
For further details on Korea’s clinical trial environment and updates, visit Intoinworld’s Clinical Trial Information page. Keeping abreast of the latest regulatory changes and industry trends – including the progress in ADC research – is vital for devising an effective development strategy.
Planning a new clinical trial? Contact Intoinworld today for a free project consultation and quote, and let our experts help you navigate the Korean clinical trial landscape.
Stay informed on the latest pharma and biotech clinical trial trends. Subscribe to Intoinworld’s newsletter for regular insights and updates delivered to your inbox.
FAQ
Q1. What are antibody-drug conjugates (ADCs) and why are they important?
A1. ADCs are targeted cancer medicines composed of an antibody linked to a potent anticancer drug. They work by specifically homing in on cancer cells (via the antibody) and then releasing the toxic drug payload inside those cells. This targeted approach can reduce side effects compared to traditional chemotherapy, as the drug is designed to mainly affect tumor tissue.
ADCs are important because they combine the precision of immunotherapy with the power of chemotherapy, offering new hope for treating cancers that are hard to target with conventional therapies.
Q2. Why conduct ADC clinical trials in South Korea?
A2. South Korea offers a conducive environment for cutting-edge trials like ADCs. The country’s regulatory agency has implemented fast-track approval processes and encourages innovation, which can speed up trial initiation. Korea also boasts a high-quality medical infrastructure with many top hospitals and experienced investigators concentrated in a relatively small area, making patient recruitment efficient. In fact, Seoul is one of the world’s most active cities for clinical research.
Data from trials conducted in Korea are respected globally due to the country’s adherence to ICH-GCP standards, meaning results from Korean ADC trials can support international drug approval efforts. Additionally, trial costs in Korea are often lower than in the US or EU, offering cost-effectiveness without compromising quality.
Q3. What is the role of a CRO in clinical trials, and how can it help in ADC drug development?
A3. A contract research organization (CRO) plans and executes clinical trials on behalf of the drug developer. For ADC development, a CRO will handle regulatory submissions (e.g. preparing the IND for MFDS or FDA), coordinate with hospital sites and principal investigators to run the trial, monitor patient safety and data quality, and analyze results. In short, the CRO takes care of the operational logistics and regulatory compliance, allowing the sponsoring company to focus on the science.
When working in a specialized area like ADCs, a CRO with relevant oncology experience can be especially valuable, ensuring that complex protocols are properly implemented and that any challenges (such as managing novel toxicity profiles or biomarker assays) are expertly handled. Using a CRO — particularly a local one in Korea — can significantly smooth out the process of launching and completing an ADC trial, overcoming language barriers and leveraging established relationships with local trial sites.