Why Korea Has Become a Strategic Global Clinical Trial Hub
Over the past few years, South Korea has evolved from a regional Asian clinical trial market into one of the world’s most strategic hubs for innovative drug development. Particularly in oncology, antibody-drug conjugates (ADC), cell and gene therapy (CGT), immunotherapy, and rare disease research, an increasing number of global pharmaceutical companies and biotech firms are incorporating Korea into their core clinical development strategies.
For many international sponsors, Korea’s biggest strengths come from several key factors, including large tertiary hospital systems, high-quality clinical data, experienced investigators, highly digitized healthcare infrastructure, and concentrated patient populations. At the same time, the Ministry of Food and Drug Safety (MFDS) continues to expand support policies for innovative global drug development, further strengthening Korea’s competitiveness in the international clinical trial landscape.
However, many global companies entering Korea quickly realize an important reality: the most complex part of running clinical trials in Korea is often not the regulatory approval itself, but how Korean hospitals actually operate global clinical studies internally.
How Korean Hospital Operations Differ From Western Markets
Many overseas sponsors have extensive experience in the US and European markets, so they naturally assume Korean hospital workflows function in a similar way. In reality, Korean tertiary hospitals operate under highly structured and complex internal systems, and these operational dynamics often become the true determining factor behind study startup timelines, site activation speed, and patient enrollment performance.
Korean hospitals are not simply investigator-driven environments. Once an international clinical trial enters a Korean hospital, the study typically involves multiple internal departments simultaneously, including clinical trial centers, institutional review boards (IRBs), legal departments, finance teams, pharmacies, clinical research coordinators (CRCs), laboratory departments, and hospital administration teams.
In other words, successful study startup requires far more than investigator agreement alone — it depends on coordination across multiple hospital functions.
The Real Korea Clinical Trial Startup Process
Many international sponsors initially view Korean clinical trials as a straightforward process where patient enrollment begins immediately after regulatory approval. In practice, however, Korea’s real startup process often includes hospital contract negotiation, IRB review, vendor coordination, internal hospital preparation, pharmacy setup, and site staff alignment before enrollment can actually begin.
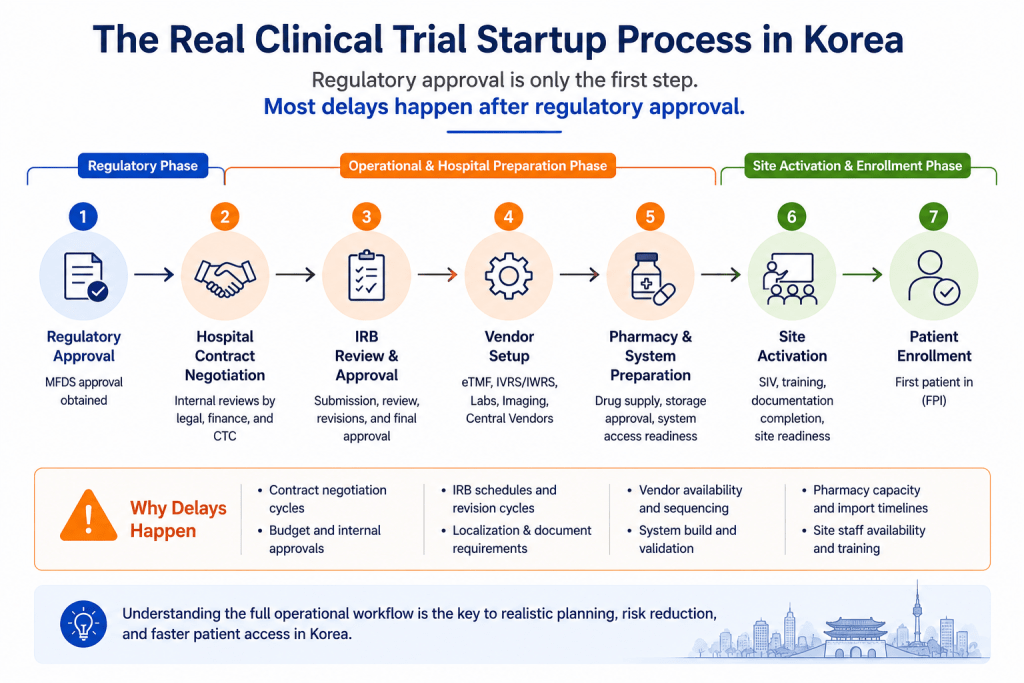
The biggest timeline risks frequently occur after approval rather than before it.
For example, even after receiving MFDS approval, studies may still face delays because contracts are incomplete, budgets are not finalized, IRB documents require revisions, pharmacy workflows are pending, vendor timelines are misaligned, or site staff preparation has not yet been completed.
For many sponsors entering Korea for the first time, the most underestimated challenge is the complexity of hospital operations.
Why Hospital Contract Negotiation Often Takes Longer in Korea
In particular, the administrative structure inside large Korean hospitals is often far more complicated than international sponsors initially expect.
Hospital contract negotiation is a common example. Many overseas companies assume contracts only require simple legal review. In Korean hospitals, however, contracts often go through multiple layers of internal review involving clinical trial centers, legal departments, finance teams, and budget approval committees.
These reviews are typically not conducted simultaneously, which can significantly extend negotiation timelines.
As a result, what appears to be a routine contract process can become one of the most unpredictable bottlenecks in overall trial startup.
Why Korean IRBs Frequently Become Timeline Bottlenecks
Large Korean hospitals typically operate under fixed IRB review cycles and strict submission requirements. Different hospitals may have different submission windows, review frequencies, formatting requirements, and patient document expectations.
Missing a submission deadline can delay the entire project timeline by several weeks.
At the same time, Korean hospitals place significant emphasis on localization quality for patient-facing documents.
Many overseas sponsors assume that simply translating English documents into Korean is sufficient. In reality, Korean hospitals evaluate whether medical terminology aligns with Korean clinical standards, whether patients can easily understand the language, whether document formatting follows hospital expectations, and whether wording sounds natural in Korean clinical practice.
As a result, even small wording inconsistencies may trigger IRB revision requests.
Many global studies experience startup delays in Korea not because of regulatory issues, but because of localization challenges.
Why Large Korean Hospitals Are Not Always Faster
As Korea continues to expand rapidly in oncology, ADC, and CGT research, competition inside major Korean hospitals is also increasing significantly.
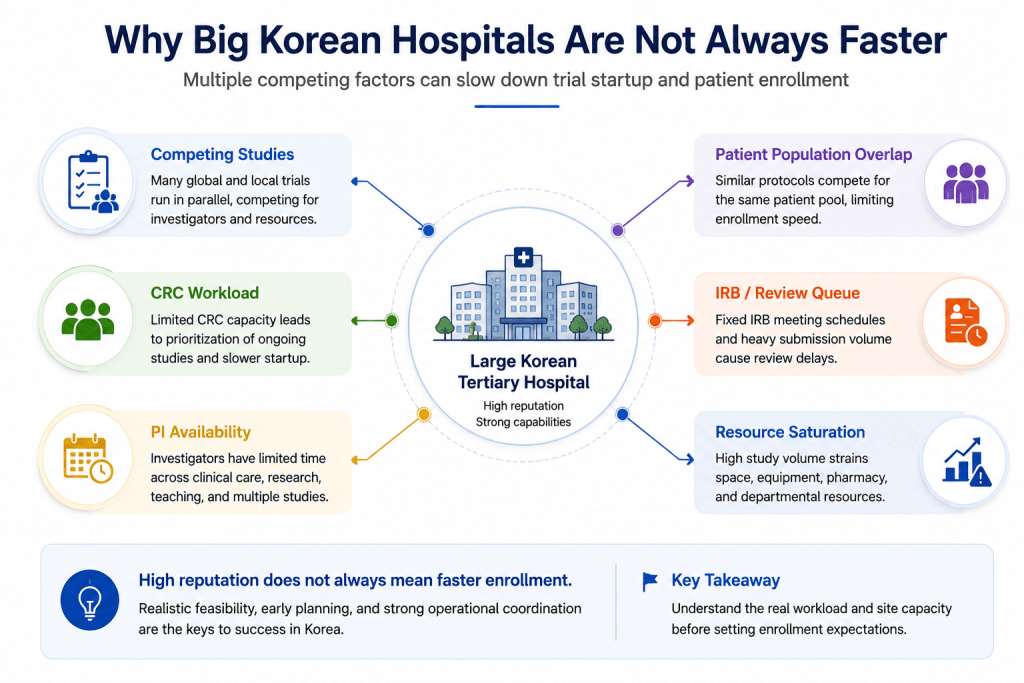
Many global sponsors assume that selecting a top Korean hospital automatically guarantees fast patient enrollment. In reality, large hospitals are often simultaneously managing numerous international studies.
Particularly in oncology, a single hospital may already be running multiple global Phase III studies, ADC programs, immunotherapy trials, and rare disease protocols at the same time.
As a result, even highly experienced hospitals may face slower startup or enrollment timelines due to CRC workload saturation, limited investigator availability, overlapping patient pools, and increasing competition between protocols.
This means that hospital reputation alone is not the most important factor. What matters more is current site workload, competing studies, realistic patient accessibility, and whether feasibility assumptions are truly achievable.
Many enrollment delays occur not because hospitals lack capability, but because feasibility expectations were overly optimistic from the beginning.
Why Operational Coordination Is Becoming More Important in Korea
At the same time, Korean hospitals are placing increasing importance on the operational capabilities of sponsors and CRO partners.
In the past, many overseas companies viewed CROs primarily as regulatory support providers. In Korea, however, the most valuable local teams are often those capable of handling hospital communication, startup coordination, timeline management, vendor synchronization, and operational risk mitigation.
This is because the most difficult part of Korean clinical trials is often not regulation itself, but cross-functional operational coordination.
Particularly in multinational studies involving multiple vendors and stakeholders, delays in one area can easily affect the entire startup process.
For example, contract delays may postpone IRB submission, which can then delay vendor setup, site activation, and ultimately patient enrollment timelines.
This highly interconnected operational structure is one of the defining characteristics of Korean clinical trials.
Why Understanding Korean Hospital Operations Matters More Than Ever
As a result, more global sponsors are beginning to realize that success in Korea depends not only on regulatory approval speed, but on overall operational execution capability.
Companies that truly understand how Korean hospitals handle global clinical trials are generally better positioned to reduce startup delays, improve hospital collaboration, minimize operational risk, and strengthen overall study execution efficiency.
Because in Korea, the most complex part of clinical trials is often not regulation — it is operational management.
Korea remains one of the world’s most strategically important clinical trial markets. Particularly in oncology, ADC, CGT, and early-phase research, Korean hospital systems continue to offer exceptional research infrastructure, experienced investigators, and globally competitive data quality.
At the same time, increasing global trial volume is creating greater operational complexity and resource competition within Korean hospitals.
For international sponsors, the key challenge is no longer simply how to obtain Korean regulatory approval, but how to truly understand how Korean hospitals operate global clinical trials.
Because in Korea, operational coordination — not regulation alone — is often what determines clinical trial execution efficiency.
Planning a Clinical Trial in Korea?
As Korea continues to strengthen its strategic role in oncology, ADC, CGT, and innovative global drug development, more international sponsors are reassessing their Korea clinical trial strategies.
However, the real challenges of Korean clinical trials often go beyond regulatory approval. Hospital operational workflows, startup coordination, IRB management, contract negotiation, localization execution, and multi-stakeholder coordination are becoming increasingly important factors in study success.
If you are evaluating Korea clinical trial feasibility, startup strategy, oncology or ADC execution environments, or Korean hospital selection strategies, the Intoinworld team can help support your planning process.
We assist global sponsors in assessing Korea startup strategy, operational feasibility, and local execution planning for more efficient clinical trial delivery.
👉 Contact us to discuss your Korea clinical trial strategy.

