The Structural Evolution of the Next-Generation Cell Therapy Market
Over the past decade, chimeric antigen receptor T-cell (CAR-T) therapies have fundamentally transformed the landscape of oncology, achieving unprecedented complete remission rates in patients with refractory hematological malignancies. However, the conventional ex vivo manufacturing model presents severe structural limitations. This process requires harvesting the patient’s T cells via leukapheresis, transporting them to a centralized GMP facility, genetically engineering them to express the CAR, expanding the cell population, and finally reinfusing them into the patient. This multi-step workflow generates a vein-to-vein time of several weeks, during which patients with aggressive disease progression may succumb before receiving treatment. Furthermore, the astronomical cost of goods sold (COGS) and the strict requirement for harsh lymphodepleting preconditioning chemotherapy have severely constrained the global scalability of these life-saving therapies.
As of 2026, the biopharmaceutical industry is undergoing a massive paradigm shift with the rapid clinical translation of “in vivo CAR-T” technologies. By utilizing targeted lipid nanoparticles (LNPs) or engineered viral vectors administered directly via intravenous infusion, in vivo CAR-T therapies bypass the ex vivo manufacturing bottleneck entirely, reprogramming the patient’s endogenous T cells directly within the body. This transition from a highly bespoke, individualized cellular manufacturing service to an off-the-shelf, scalable pharmaceutical product fundamentally alters the therapeutic risk profile. Consequently, the industry is witnessing an urgent need to redesign the protocols and frameworks governing modern Clinical Trials. This comprehensive report analyzes the aggressive M&A trends of major pharmaceutical companies, the emerging challenges in Clinical Trials protocol design, the latest regulatory flexibilities introduced by the US FDA, and the geopolitical shifts positioning South Korea as the premier destination for executing advanced global Clinical Trials.
Scientific Foundations and Platform Classifications of In Vivo T-Cell Engineering
The clinical viability of in vivo CAR-T therapy depends entirely on the precision and targeting efficiency of the gene delivery vehicle. The payload must safely bypass the innate immune system, avoid sequestration in non-target organs such as the liver, and exclusively transduce the targeted T-cell populations. Currently, the delivery platforms advancing into human Clinical Trials are broadly classified into two categories: viral vectors and non-viral vectors.
Viral Vector Platforms: Lentivirus and iGPS Technology
Lentiviral vectors have long been the gold standard for ex vivo CAR-T engineering due to their high transduction efficiency and ability to stably integrate genetic material into the host genome. However, systemic administration of traditional lentiviruses poses a risk of off-target transduction and insertional mutagenesis.
To resolve this, companies like Kelonia Therapeutics have developed advanced in vivo gene placement systems (iGPS®). By modifying the viral envelope glycoproteins to eliminate natural tropism and incorporating specific targeting ligands (such as anti-CD8 antibodies), these engineered vectors selectively bind to and transduce T cells directly in the bloodstream. Because the viral vector facilitates permanent genomic integration, this platform provides highly durable and sustained CAR expression, making it exceptionally well-suited for oncology Clinical Trials targeting aggressive malignancies like relapsed/refractory multiple myeloma.
Non-Viral Vector Platforms: LNP and circRNA Technology
Conversely, lipid nanoparticle (LNP) technologies represent a non-viral, non-integrating approach to in vivo T-cell engineering. Leading innovators such as Orna Therapeutics utilize proprietary circular RNA (circRNA) packaged within specialized LNPs to deliver the CAR genetic code.
Unlike linear mRNA, circRNA exhibits superior intracellular stability and drives higher levels of protein expression over an extended duration. Because this platform relies on transient episomal expression rather than permanent genomic integration, the resulting CAR-T cells naturally decay after a few weeks. This transient therapeutic window is highly advantageous for autoimmune disease Clinical Trials, such as those for Systemic Lupus Erythematosus (SLE), where the clinical objective is a temporary “immune reset” via the eradication of pathogenic B cells, rather than lifelong CAR-T persistence.
| Evaluation Metric | Traditional Ex Vivo CAR-T | Viral In Vivo CAR-T (e.g., Lentivirus) | Non-Viral In Vivo CAR-T (e.g., LNP/circRNA) |
| Manufacturing Model | Decentralized or centralized patient-specific manufacturing | Mass-produced, scalable off-the-shelf pharmaceutical | Mass-produced, scalable off-the-shelf pharmaceutical |
| Treatment Timeline | 3 to 6 weeks (apheresis to reinfusion) | Immediate (intravenous injection) | Immediate (intravenous injection) |
| Lymphodepletion | Mandatory for engraftment | Generally not required | Not required |
| Expression Profile | Permanent (Genomic integration) | Permanent (Genomic integration) | Transient (Cytoplasmic translation, decays over weeks) |
| Clinical Trials Risks | Severe CRS, ICANS, manufacturing failure | Insertional mutagenesis, ADA development | Off-target organ accumulation (liver), repeat dosing ADA |
| Target Indications | Hematological malignancies | Hematological and solid tumors | Autoimmune diseases (immune reset) and oncology |
Table 1: Comparative analysis of traditional ex vivo CAR-T versus emerging in vivo CAR-T modalities in the context of modern Clinical Trials.
2026 Big Pharma M&A Trends: Eli Lilly’s Comprehensive Pipeline Expansion
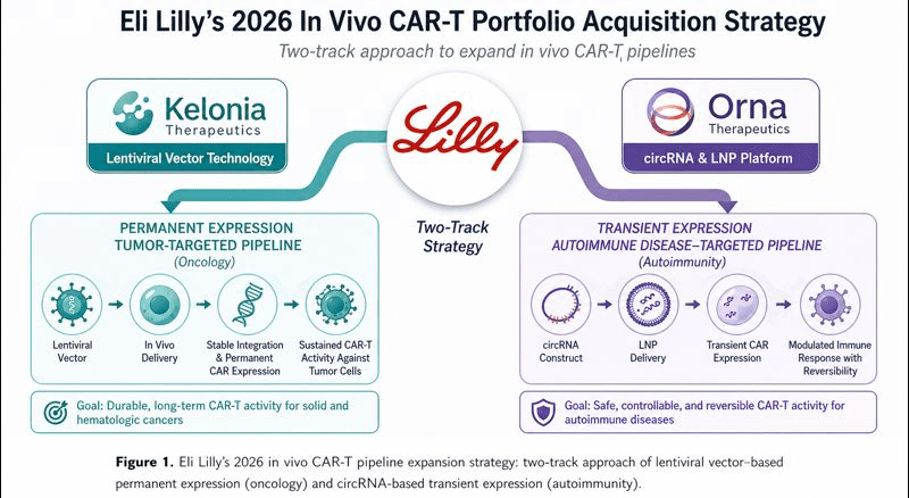
As in vivo CAR-T platforms successfully transition from preclinical proof-of-concept to active human Clinical Trials, multinational pharmaceutical companies have initiated a highly aggressive wave of mergers and acquisitions to secure next-generation capabilities. Fueled by unprecedented cash flows from its GLP-1 metabolic franchise, Eli Lilly emerged as the dominant force in 2026, executing two monumental acquisitions to cement its leadership in genetic medicines.
Kelonia Therapeutics: A $7 Billion Acquisition
In April 2026, Eli Lilly announced a definitive agreement to acquire Kelonia Therapeutics for up to $7 billion, comprising a $3.25 billion upfront payment and $3.75 billion in subsequent milestones.3 Kelonia’s lead asset, KLN-1010, is a first-in-class in vivo CAR-T therapy currently undergoing Phase 1 Clinical Trials for relapsed/refractory multiple myeloma.
The early clinical data has been extraordinary. Administered as a single intravenous infusion without any prior lymphodepleting chemotherapy, KLN-1010 successfully generated robust in vivo CAR-T expansion. Initial cohorts demonstrated a 100% minimal residual disease (MRD) negative response rate, accompanied by a highly favorable safety profile devoid of severe cytokine release syndrome (CRS) or neurotoxicity.3 This acquisition grants Lilly a validated viral platform capable of dominating future oncology Clinical Trials.
Orna Therapeutics: A $2.4 Billion Acquisition
To balance its permanent-expression oncology portfolio with a transient-expression platform tailored for immunology, Eli Lilly also acquired Orna Therapeutics in February 2026 for $2.4 billion.5 Orna’s lead candidate, ORN-252, is an in vivo CAR-T therapy targeting CD19, meticulously designed to eradicate disease-driving B cells.
As ORN-252 advances into Phase 1 Clinical Trials for autoimmune diseases, Lilly’s Immunology leadership emphasized that the astronomical costs and complex logistics of ex vivo cell therapies are insurmountable barriers for broader autoimmune patient populations. Orna’s circRNA/LNP platform elegantly solves this challenge, allowing Lilly to aggressively expand its autoimmune Clinical Trials footprint globally.
Industry Ripple Effects and Pipeline Acceleration
Lilly is not alone in this feeding frenzy. The broader industry has witnessed AbbVie acquiring Capstan Therapeutics for $2.1 billion, Bristol Myers Squibb absorbing Orbital Therapeutics for $1.5 billion, and Gilead/Kite securing Interius BioTherapeutics. As these massive pharmaceutical sponsors integrate these platforms, the global Clinical Trials ecosystem is bracing for an unprecedented influx of highly complex, genetically engineered therapies targeting both rare and prevalent diseases.
Challenges and Strategies in Next-Generation Clinical Trials Protocol Design
Administering a gene therapy vector directly into a patient’s bloodstream to manufacture living cellular drugs in situ completely disrupts traditional pharmacokinetic and pharmacodynamic models. Consequently, the architecture of Clinical Trials is undergoing a profound transformation to ensure patient safety and scientific rigor.
Biomarker-Driven Dosing and PK/PD Modeling
In traditional ex vivo Clinical Trials, investigators administer a precisely quantified number of viable, engineered CAR-T cells. In sharp contrast, in vivo CAR-T Clinical Trials administer a dose of vector particles (e.g., viral genomes or LNP mass). The subsequent in vivo transduction efficiency and the peak expansion of the newly generated CAR-T cells depend heavily on the patient’s unique immune baseline. Therefore, modern Clinical Trials must incorporate sophisticated liquid biopsy monitoring, utilizing high-resolution multiparameter flow cytometry and next-generation sequencing to dynamically track vector biodistribution, T-cell transduction rates, and real-time CAR protein expression.
Controlling Safety: On-Target Off-Tumor Toxicity and Immunogenicity
The systemic administration of gene delivery vehicles introduces unique safety considerations that must be meticulously managed during Clinical Trials.
- On-Target Off-Tumor Toxicity: If the delivery vector erroneously transduces non-target cells (such as macrophages or regulatory T cells), it can precipitate severe off-tumor toxicities or paradoxical immune suppression. Rigorous biomarker screening and tissue biopsies are essential in early-phase Clinical Trials to verify vector tropism.
- Vector Immunogenicity: Patients may possess pre-existing neutralizing antibodies against viral capsids or LNP lipid components. Furthermore, repeated dosing—often necessary for mRNA platforms—can trigger anti-drug antibodies (ADA). Managing this requires integrating precision immunomodulatory regimens into the Clinical Trials protocol.
- Safety Switches: To mitigate the risks of unconstrained in vivo proliferation and life-threatening CRS, next-generation constructs routinely incorporate small-molecule-gated “safety switches.” These engineered logic gates allow investigators conducting Clinical Trials to rapidly halt CAR-T activity or induce cellular apoptosis if severe toxicities manifest.
15-Year Long-Term Follow-Up (LTFU) Requirements
Because integrating vectors (like lentiviruses) carry an inherent risk of insertional mutagenesis and the theoretical potential for secondary primary malignancies (SPMs), global regulatory bodies strictly mandate exhaustive long-term surveillance. Sponsors of in vivo CAR-T Clinical Trials must establish robust data registry infrastructures capable of tracking patients for up to 15 years post-infusion.9 This demands a seamless integration of clinical data management systems that can maintain data integrity long after the primary Clinical Trials endpoint has been met.
Evolving Global Regulatory Frameworks: 2026 US FDA CMC Flexibility
To support the explosive growth of complex cellular and genetic medicines, regulatory authorities are adapting their oversight mechanisms. On January 11, 2026, the US FDA’s Center for Biologics Evaluation and Research (CBER) issued groundbreaking guidance offering unprecedented regulatory flexibility regarding Chemistry, Manufacturing, and Controls (CMC) requirements for cell and gene therapies.
FDA Commissioner Dr. Marty Makary stated that these common-sense reforms are explicitly designed to dismantle administrative barriers and foster rapid innovation.11 Under this new framework, sponsors conducting Phase 2 and Phase 3 Clinical Trials are no longer expected to strictly comply with the onerous commercial manufacturing standards of 21 CFR part 211 (exempted under 21 CFR 210.2(c)). Furthermore, if sponsors implement minor manufacturing process alterations during late-stage Clinical Trials, the FDA will now accept streamlined comparability data rather than demanding massive, time-consuming re-validation studies. This paradigm shift drastically reduces the timeline and capital required to progress an in vivo CAR-T asset from first-in-human Clinical Trials to commercial licensure.
| Regulatory Domain | Traditional FDA CMC Expectations | 2026 FDA CMC Flexibility for CGT Clinical Trials |
| Phase 2/3 Manufacturing | Full compliance with commercial 21 CFR part 211 | Broad exemptions granted under 21 CFR 210.2(c) |
| Product Release Criteria | Strict, finalized specifications required early | Permissive acceptance criteria allowed during Clinical Trials |
| Process Changes | Onerous and highly restrictive comparability data | Minor changes approved rapidly with streamlined comparability |
| Validation Requirements | Mandatory submission of three (3) PPQ lots | Requirement for three PPQ lots waived, embracing lifecycle evolution |
Table 2: Summary of the FDA’s 2026 CMC regulatory flexibility initiatives designed to accelerate advanced Clinical Trials.
The BIOSECURE Act and the Geopolitical Realignment of Clinical Trials Supply Chains
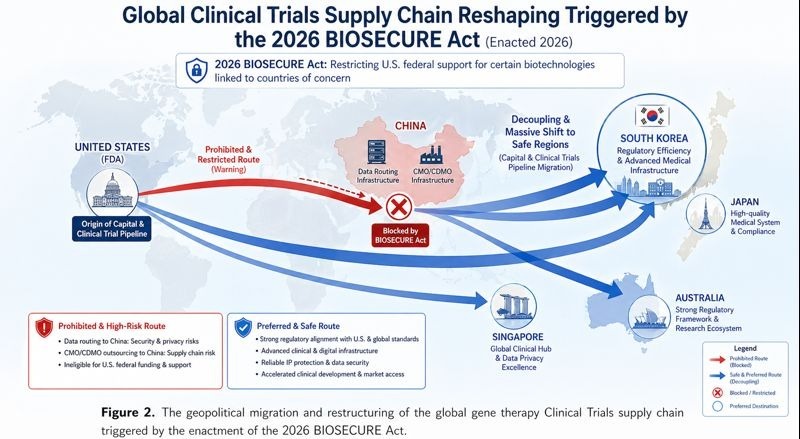
While technological and regulatory advancements are propelling the industry forward, a massive geopolitical disruption is simultaneously reshaping the global landscape of Clinical Trials. The BIOSECURE Act, signed into US law and fully actionable in 2026, prohibits organizations receiving federal funding from utilizing biotechnology equipment, services, or data platforms provided by designated “biotechnology companies of concern” linked to foreign adversaries.
Congressional investigations revealed concerns regarding certain Chinese military-affiliated medical institutions allegedly misappropriating multiomic and genomic data from Western pharmaceutical Clinical Trials. Because in vivo CAR-T Clinical Trials heavily depend on intensive genomic sequencing, patient immune profiling, and high-resolution biomarker tracking, global sponsors are actively terminating contracts with Chinese CDMOs and clinical research organizations. This legislative mandate has triggered an urgent, massive decoupling of the global Clinical Trials supply chain, forcing sponsors to rapidly relocate their highly sensitive genetic medicine portfolios to secure, US-allied clinical hubs.
Asia-Pacific Hub: South Korea’s Clinical Trials Competitiveness and Global CRO Strategy
As the BIOSECURE Act forces a rapid geographic realignment of advanced R&D, South Korea has firmly established itself as the premier alternative destination for next-generation Clinical Trials. Boasting an exceptional regulatory environment, highly concentrated medical infrastructure, and impeccable data integrity standards, South Korea consistently ranks among the top 4 global hubs for Clinical Trials.
Unmatched Regulatory Efficiency and Start-Up Timelines
In the highly competitive race to validate in vivo CAR-T therapies, time-to-market is the ultimate currency. South Korea’s Ministry of Food and Drug Safety (MFDS) operates a highly efficient regulatory apparatus that allows the IND administrative review and the hospital-level Institutional Review Board (IRB) ethics review to be conducted in parallel. This streamlined process results in an astonishing average start-up timeline of just 6 to 8 weeks to achieve First-Patient-In (FPI). For a comprehensive analysis of early-phase regulatory strategies, global sponsors can explore https://intoinworld.com/korea-clinical-trials-phase-i-ii-strategy/ to understand how to optimize their timelines.
Top-Tier Infrastructure and High Patient Density
Seoul remains the undisputed global capital for Clinical Trials activity, supported by a dense network of massive tertiary academic hospitals. Institutions like Seoul National University Hospital (SNUH) have executed over 945 early-phase oncology Clinical Trials within a brief five-year window. These mega-hospitals are equipped with proprietary AI-driven clinical decision support systems (such as KMed.AI) and in-house GMP facilities perfectly suited for translational research. For complex in vivo CAR-T Clinical Trials that require 24/7 ICU access and rapid intervention protocols to manage potential neurotoxicity or CRS, South Korea’s clinical infrastructure offers unparalleled safety and operational execution.
Integrated Clinical Trials Operating System via Global CROs
Navigating this sophisticated landscape requires the expertise of specialized global Contract Research Organizations (CROs) operating within the region. Modern CROs function as a comprehensive “Operating System” for Clinical Trials, seamlessly coordinating cross-border logistics, navigating local MFDS regulations, and ensuring rigorous ICH GCP compliance. Korean clinical data is universally accepted by the FDA and EMA for pivotal global submissions, ensuring that sponsors maximize the ROI of their developmental programs. To discover why top pharmaceutical companies are making this transition, you can read the detailed guide at https://intoinworld.com/south-korea-clinical-trials-powerhouse/ for unparalleled insights.
| Key Performance Indicator | South Korea’s Clinical Trials Metrics | Strategic Advantage for Global Sponsors |
| Trial Start-Up Timeline | 6 to 8 weeks (Parallel IND/IRB Review) | Months faster than competing APAC regions; accelerates time-to-market |
| Global Ranking | Top 4 globally for overall trial activity | Proven ecosystem with rapid and reliable patient recruitment |
| Infrastructure Density | >220 MFDS-certified mega-hospitals | Highly efficient execution of complex, multi-center protocols |
| Data Integrity & Acceptance | Full ICH GCP compliance | Data is unconditionally accepted by the FDA and EMA for BLA submissions |
Table 3: Strategic metrics positioning South Korea as the premier destination for advanced genetic medicine Clinical Trials following the 2026 supply chain realignment.
Conclusion: Finalizing the Genetic Medicine Revolution
The advent of in vivo CAR-T technology represents the final frontier in cellular immunotherapy, systematically dismantling the logistical, financial, and biological barriers that have long plagued ex vivo manufacturing. Eli Lilly’s aggressive $9.4 billion combined acquisition of Kelonia Therapeutics and Orna Therapeutics in early 2026 definitively signals that the pharmaceutical industry is aggressively pivoting toward in situ cellular reprogramming for both oncology and autoimmune disease indications.
However, successfully commercializing these disruptive modalities requires navigating a labyrinth of novel challenges within Clinical Trials. From establishing sophisticated biomarker-driven dosing protocols to managing 15-year longitudinal safety registries, the operational demands on clinical sponsors have never been higher. Compounded by the FDA’s new flexible CMC guidelines and the geopolitical decoupling mandated by the BIOSECURE Act, executing global Clinical Trials today requires unparalleled strategic foresight. For visionary biopharmaceutical sponsors, leveraging the exceptional speed, clinical infrastructure, and uncompromised data integrity of South Korea is no longer merely an option—it is an absolute imperative to lead the global genetic medicine revolution.
Are you ready to optimize your In Vivo CAR-T Clinical Trials design?
Developing next-generation cell and gene therapies requires navigating complex biomarker strategies, stringent long-term follow-up protocols, and rapidly shifting global regulations. Partner with our world-class CRO experts in South Korea to experience unparalleled execution speed—achieving trial start-up in just 6 to 8 weeks. Maximize your clinical ROI with FDA-compliant data integrity today.
Stay ahead of the curve in Global Clinical Trials Intelligence!
Don’t miss the latest updates on FDA CMC regulatory flexibilities, Big Pharma M&A dynamics, and exclusive insights into the Asia-Pacific clinical landscape. Subscribe to our premium insights to receive expert analyses delivered directly to your inbox.
Frequently Asked Questions (FAQs)
Q1. How do in vivo CAR-T Clinical Trials fundamentally differ from traditional ex vivo CAR-T studies?
A1. In traditional ex vivo Clinical Trials, a patient’s T cells must be extracted, transported to a lab, genetically modified, expanded, and then reinfused—a costly process taking several weeks. In vivo CAR-T Clinical Trials bypass this entirely by administering a specialized gene delivery vector (like an LNP or lentivirus) directly into the patient’s bloodstream, programming their immune cells on the spot. This eliminates the need for complex manufacturing logistics and harsh preconditioning chemotherapy, drastically accelerating treatment timelines.
Q2. What is the strategic significance of Eli Lilly acquiring both Kelonia and Orna Therapeutics for their future Clinical Trials?
A2. Eli Lilly strategically acquired both companies to dominate two distinct therapeutic areas in their future Clinical Trials. Kelonia (acquired for up to $7 billion) utilizes lentiviral vectors that permanently integrate CAR genes into T cells, which is highly effective for durable cancer therapies like multiple myeloma. Conversely, Orna Therapeutics ($2.4 billion) uses circular RNA within LNPs to drive transient CAR expression, making it exceptionally safe and effective for immune-reset strategies in autoimmune disease Clinical Trials.
Q3. What are the most critical safety parameters monitored during in vivo CAR-T Clinical Trials?
A3. Because gene-editing vectors are administered systemically, investigators conducting Clinical Trials must rigorously monitor for “on-target off-tumor” toxicities and unintended vector accumulation in organs like the liver.8 Furthermore, managing the immunogenicity of the delivery vehicle (such as anti-drug antibodies) and mitigating the risk of insertional mutagenesis from viral vectors necessitates the integration of safety switches and mandatory 15-year long-term follow-up (LTFU) observation protocols.
Q4. How does the 2026 FDA CMC guidance impact the timeline for advanced gene therapy Clinical Trials?
A4. The FDA’s January 2026 guidance introduces significant regulatory flexibility to accelerate innovation. For sponsors conducting Phase 2 and Phase 3 Clinical Trials, the FDA has relaxed strict compliance with commercial 21 CFR part 211 manufacturing standards.10 Additionally, minor manufacturing changes during late-stage Clinical Trials can now be approved using streamlined comparability data, drastically reducing the time and financial burden required to reach commercialization.
Q5. Why is South Korea emerging as the preferred hub for global Clinical Trials following the BIOSECURE Act?
A5. The US BIOSECURE Act severely restricts partnerships with specific foreign entities due to data security concerns, forcing sponsors to relocate their highly sensitive genetic Clinical Trials out of China. South Korea has become the premier alternative because it offers a highly advanced medical infrastructure, uncompromising ICH GCP data integrity (fully accepted by the FDA), and an incredibly efficient parallel regulatory review process that enables sponsors to launch Clinical Trials in just 6 to 8 weeks.



