As we enter 2026, Cell and Gene Therapy (CGT) has transcended the boundaries of specialized laboratory research to become the most critical frontline in the global pharmaceutical industry’s quest for market dominance. South Korea, bolstered by its highly centralized medical infrastructure, advanced digital clinical environment, and strategic government mandates for cutting-edge biotechnology, has emerged as the premier strategic hub for CGT clinical trials in the Asia-Pacific region.
However, the inherent heterogeneity of CGT products, the complexity of Chemistry, Manufacturing, and Controls (CMC), and the potential for long-term safety risks have prompted the Ministry of Food and Drug Safety (MFDS) to implement a significantly more granular and rigorous regulatory framework in 2026. For global sponsors, deciphering the scientific logic behind these new mandates is not merely a matter of compliance—it is a strategic necessity that determines the commercial survival of their developmental pipelines.
The Paradigm Shift in Supervision: Transitioning to Total Product Life Cycle (TPLC) Compliance
Under the traditional regulatory paradigms of the past decade, the collaboration between Contract Research Organizations (CROs) and Sponsors was often focused on a “static” submission—packaging data for the singular moment of an Investigational New Drug (IND) application. However, within the 2026 framework, the MFDS has explicitly expanded its oversight to the Total Product Life Cycle (TPLC). This signifies that the regulatory body is no longer satisfied with static data snapshots at the onset of a trial. Instead, it requires Sponsors to establish a compliance ecosystem capable of dynamic, real-time monitoring. The most direct manifestation of this shift is the mandate for Long-term Follow-up (LTFU) for cell-modified products, stretching up to 15 years.
The 2026 regulations explicitly state that LTFU data can no longer rely on legacy paper-based records or isolated spreadsheets. Data must be integrated into digital platforms that strictly adhere to 21 CFR Part 11 standards and possess real-time signal detection capabilities for adverse events. This poses a massive technical hurdle for many biotech companies still in early-phase clinical development: How can they ensure that patient data, dispersed across multiple medical centers, remains integral, traceable, and secure over a span of fifteen years? This requirement is far more than a legal checkbox; it is a profound test of a CRO’s capability in digital infrastructure and long-term data stewardship.
Bridging the Technical Fissure Between CMC and Clinical Pathways: The “Process is Product” Imperative
In the realm of CGT, the axiom “The Process is the Product” remains the ultimate truth. Yet, in practical execution, the most frequent point of failure for Sponsors is the severe disconnection between CMC data and clinical reality. In 2026, the MFDS has introduced meticulously detailed standards for Viral Vector quality control, emphasizing the precise characterization of empty capsids and the specific evaluation of their impact on human immunogenicity and safety. Many biotechs, when filing cross-border applications, tend to recycle data packages approved by the US FDA or EMA, neglecting the unique technical indicators required by the MFDS for “Advanced Biologics.”
For instance, the MFDS now demands more dynamic validation data regarding cell expansion fold, purification efficiency, and batch consistency. If a CRO lacks the technical depth to grasp these CMC nuances and fails to conduct “stress tests” prior to submission to anticipate the regulator’s concerns, a pipeline can easily be derailed by indefinite Requests for Information (RFIs). For innovation-driven firms where time is the most expensive commodity, such delays can be commercially devastating. Intoinworld bridges this gap by aligning manufacturing stability with regulatory expectations through rigorous pre-submission technical audits.
Digital Clinical Trials (DCT) as a Formal Pathway for Precision Recruitment
The core bottleneck of CGT trials has historically been the extreme difficulty of patient recruitment, particularly for rare diseases or specific genetic mutations. In 2026, South Korea formally integrated Decentralized Clinical Trials (DCT) into the legitimate regulatory pathway for CGT, offering Sponsors a new mechanism to transcend geographical limitations. By utilizing electronic Informed Consent (eConsent), home-based sampling, and wearable physiological monitoring, Sponsors can expand their patient pool across the entire nation and even manage Asia-Pacific recruitment remotely.
However, the implementation of DCT is not merely about switching software tools. Its technical core lies in establishing Digital Standard Operating Procedures (SOPs) that comply with the newly finalized ICH E6(R3) guidelines. During audits, the MFDS focuses intensely on whether data collected in home environments possesses the same homogeneity and authenticity as that from a central lab. Intoinworld achieves this through deep data integration with the Electronic Medical Record (EMR) systems of top-tier South Korean hospitals, utilizing AI-driven pre-screening to locate rare disease patients and ensuring every digital footprint meets the rigorous field inspection standards of regulatory bodies.
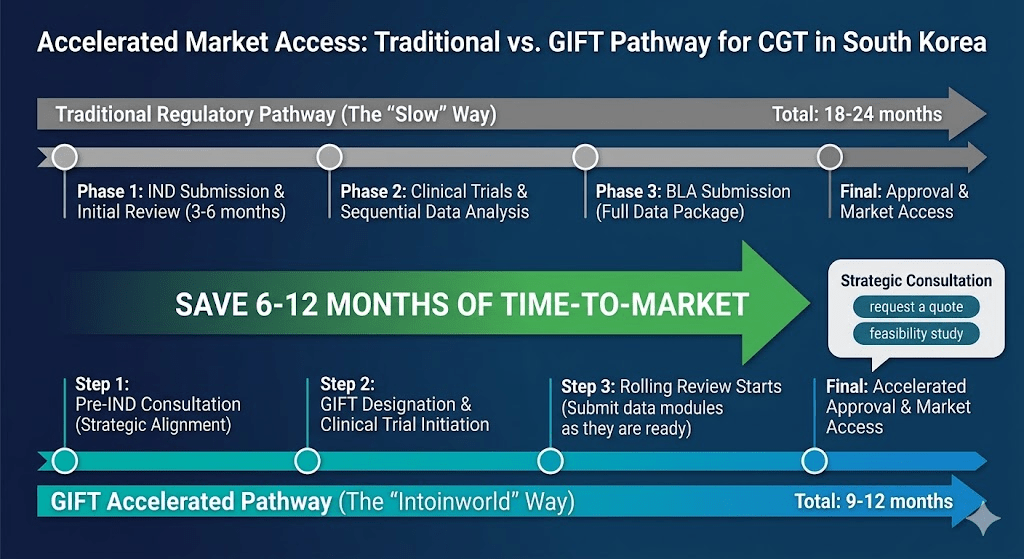
Maximizing the GIFT Accelerated Pathway and Pre-IND Strategic Communication
In a hyper-competitive market, a lead time of just three months can translate into a massive difference in commercial valuation. To achieve strategic acceleration in 2026, Sponsors must maximize the Global Innovative Products on Fast Track (GIFT) system. While GIFT offers the lucrative privilege of Rolling Review, its entry threshold is exceptionally high. It requires Sponsors to provide compelling evidence of breakthrough efficacy and a data package of the highest scientific integrity.
Before entering the formal submission phase, Pre-IND strategic communication with the MFDS has become a mandatory tactical step. Through these early dialogues, Sponsors can reach a scientific consensus with regulators on the selection of surrogate endpoints, validation methods for biomarkers, and the streamlined path for CMC documentation. This “Consensus-based Development” model effectively avoids catastrophic ideological differences during formal review. Intoinworld’s regulatory strategy team, composed of former regulatory officials, excels at aligning data packaging with the reviewer’s expectations, potentially shortening the time-to-market by 6 to 12 months.
The Scientific Battleground: Immunogenicity Monitoring and Endpoint Validation
For complex therapies such as CAR-T, TCR-T, or AAV vectors, immunogenicity monitoring is a top priority for the MFDS in 2026. Regulators are no longer satisfied with simple antibody detection reports; they demand epidemiological data regarding pre-existing antibodies specifically within the South Korean population and a specific risk assessment of their impact on therapeutic safety. This requires the CRO to possess a high level of medical writing capability and scientific depth when designing trial protocols.
Furthermore, for drugs with innovative mechanisms, traditional clinical endpoints often fail to capture early biological activity. Introducing scientifically sound surrogate endpoints and proving their correlation with long-term clinical benefit is the core of an accelerated approval. This is not just about regulatory tactics; it is a battle of scientific logic. Intoinworld, through close collaboration with top Korean immunologists and clinicians, provides Sponsors with scientifically persuasive technical solutions to ensure that innovative pipelines do not stall due to regulatory caution regarding new mechanisms.
Conclusion: Locking in the Success of Innovative Pipelines in a Shifting Landscape
The 2026 South Korean CGT market is a “golden window” of opportunity for Sponsors with foresight. While the technical threshold has been raised, the resulting increase in standardization and transparency provides a clearer roadmap for those pursuing high-quality development. Success lies in choosing a CRO partner that understands not just execution, but “strategic alignment.” Intoinworld is committed to transforming complex regulatory requirements into a competitive advantage for Sponsors, securing commercial success in a rapidly evolving landscape.
FAQ: Navigating CGT Trials in South Korea
Q: How does the MFDS classify “breakthrough” CGT products for the GIFT pathway?
A: The GIFT (Global Innovative Products on Fast Track) pathway is reserved for therapies targeting life-threatening diseases with no existing treatment or those showing significant clinical improvement over current standards. In 2026, the MFDS focuses on “clinical superiority” demonstrated through robust Phase I/II data. Sponsors must provide a “Regulator-Ready” evidence package that aligns clinical outcomes with Korean-specific patient demographics.
Q: What are the specific digital requirements for the 15-year Long-term Follow-up (LTFU)?
A: Beyond 21 CFR Part 11 compliance, the MFDS now expects an “Active Monitoring” system. This means the platform must automatically flag missing data points and trigger alerts for pre-defined safety signals. Static databases are no longer sufficient; the system must support dynamic data entry from multiple decentralized sites while maintaining a strictly audited chain of custody.
Q: Can international CMC data be used directly for South Korean IND submissions?
A: While international data provides a foundation, it often requires “Localization Adjustment.” The MFDS frequently requests additional characterization data regarding batch-to-batch consistency and specific viral vector purity levels (e.g., empty/full capsid ratios) that meet localized biological standards. Pre-IND consultation is critical to identify these “Gap Requirements” early.

