Introduction: Why Korea Remains a Strategic Location for Clinical Trials
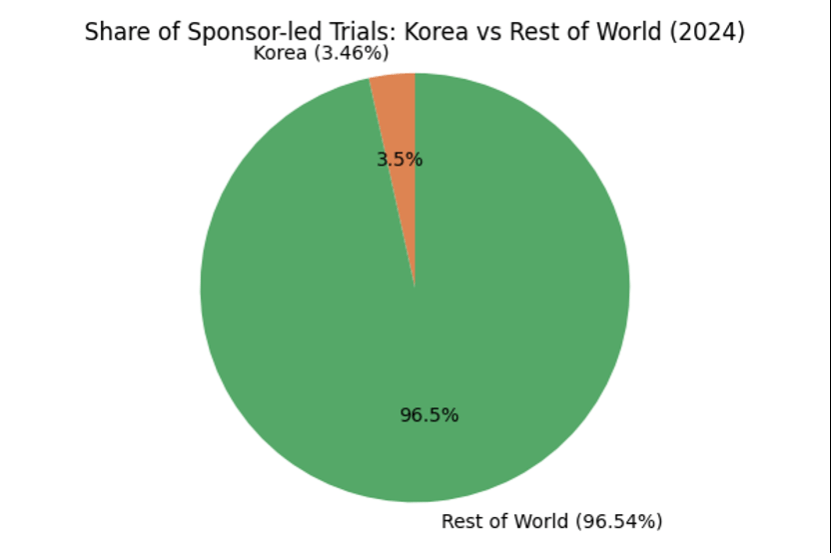
Korea’s share of global sponsor-driven Clinical Trials (3.46%) highlights its status as a major hub, ranking 6th worldwide. European pharmaceutical and biotech sponsors continue to view South Korea as a prime destination for Clinical Trials due to its fast regulatory timelines, high-quality data, and supportive infrastructure. The Ministry of Food and Drug Safety (MFDS) offers one of the world’s speediest approval processes – IND approvals are often obtained in about 4–6 weeks (30 working days). Crucially, Institutional Review Board (IRB) ethics reviews run in parallel at Korean hospitals, meaning total startup time until a study begins can be as short as 6–8 weeks. This agility far outpaces many other regions (for example, comparable approvals can take 3–4 months in China).
Beyond speed, Korea offers internationally recognized quality. As a full ICH member since 2016, Korea enforces GCP standards equivalent to the EU/US, and its data is accepted by both the EMA and FDA. Major hospitals in Seoul and across Korea provide access to a large, diverse patient pool and advanced research facilities, driving enrollment ~25% faster than the Asian average. Sponsors also benefit from cost-efficiency: conducting Clinical Trials in Korea delivers the same quality at roughly 60–70% of the cost in the United States. Thanks to strong government support (including recent MFDS reforms in 2025) and a track record of zero critical findings in FDA inspections, Korea remains a strategic location for Phase I–III trials in oncology, rare diseases, and beyond. In summary, a well-executed Clinical Trial in Korea can accelerate development timelines and provide globally credible results at lower cost – an appealing proposition for European sponsors in 2026.

Local Representation: CRO vs. Legal Entity Considerations
A key early decision for European sponsors is how to fulfill Korea’s local representation requirement. The MFDS requires foreign sponsors without a local office to appoint an in-country representative to liaise with regulators. In practice, this means European companies must either partner with a domestic CRO or establish a legal entity in Korea to act as the trial sponsor. While setting up a local subsidiary is possible (especially for large sponsors with long-term plans), most choose to work with an experienced Korea-based Contract Research Organization (CRO) or regulatory consultant. This approach satisfies the legal mandate and provides immediate local expertise. According to MFDS guidelines, a foreign sponsor without local presence is considered unable to meet all obligations, so partnering with a Korean CRO delegates those duties appropriately.
Critically, using a Korea CRO versus going it alone has strategic advantages. Local CROs are deeply familiar with MFDS processes, Korean Good Clinical Practice, and the nuances of trial conduct in Korea. They can act as the formal “local sponsor” on behalf of the European company, handling submissions, communications, and on-the-ground coordination. By contrast, if a sponsor establishes its own legal entity, it must build internal regulatory capabilities and infrastructure in Korea – a time-consuming and costly endeavor if done from scratch. Table 1 below summarizes some differences in expertise and resources when outsourcing to a CRO vs. using an in-house local entity.

Table 1. Local Sponsor Options – CRO Outsourcing vs. In-House Entity
| Aspect | CRO as Local Representative (Outsourcing) | Sponsor’s Own Local Entity (In-House) |
| Regulatory Know-how | Established MFDS liaison experience; up-to-date with Korean regulations. Handles IND/IRB prep efficiently. | Must hire/train local regulatory staff; risk of delays if unfamiliar with MFDS processes. |
| Infrastructure & SOPs | Ready systems for trial management (EDC, pharmacovigilance, translations) in place. | Need to create or adapt systems and SOPs for Korea, incurring setup time and cost. |
| Cost Structure | Variable project-based costs; avoids maintaining full in-country team. | Fixed overhead of local office and staff. Efficient only if pipeline supports continuous trials. |
| Speed & Flexibility | Quick startup due to existing local networks and processes; can rapidly allocate resources. | Slower ramp-up; limited by internal capacity and learning curve in new environment. |
| Cultural/Language | Bilingual team navigates Korean documentation, meetings, and site relationships. | In-house team may face language barriers; likely still need translators or consultants. |
Ultimately, most foreign sponsors rely on a local CRO in Korea – not only to satisfy representation requirements, but to de-risk the trial’s execution. An experienced Korean CRO will anticipate local challenges, ensure nothing “falls through the cracks” between European and Korean procedures, and keep the project on schedule. European sponsors with an existing Korean affiliate may utilize their own entity as sponsor-of-record, but even then often engage a local CRO for operational support. The 2025 MFDS regulatory reforms have increased process complexity, making local collaboration even more valuable. In summary, unless a sponsor has significant infrastructure in Korea already, partnering with a qualified Korean CRO is the recommended path to navigate regulatory submissions and trial management smoothly.
MFDS IND Approval: Key Changes and Dossier Gaps for EU-Based Sponsors
Before any Clinical Trial can start in Korea, the sponsor must obtain MFDS approval of the trial plan (Investigational New Drug application, or IND) and separate IRB approvals (covered in the next section). The MFDS IND process in 2026 remains fast and globally aligned, but European sponsors should be aware of some recent changes and potential dossier “gaps” compared to EU requirements. MFDS’s IND review timeline is officially 30 working days, similar to Europe’s CTA timelines, but in practice approval often comes faster (~4–6 weeks). Notably, since 2019 the MFDS offers pre-IND consultations: sponsors can informally discuss their plan and data with MFDS reviewers beforehand, which in some cases shortens the formal review to as little as 7 days. Utilizing this pre-submission meeting is a best practice to clarify any Korea-specific questions (e.g. need for local data) and avoid review delays. Under the 2025 regulatory reforms, the MFDS also introduced more frequent sponsor consultations and even rolling submission of dossier modules for certain applications, making the approval process more interactive. These changes signal that Korea’s regulator is keen to accelerate development timelines for innovative trials.
For European sponsors used to EMA’s processes, the content of a Korean IND application will feel familiar – it follows the ICH Common Technical Document structure
freyrsolutions.com. You’ll need to submit the clinical protocol, Investigator’s Brochure, CMC (Chemistry, Manufacturing, Control) data, nonclinical study results, prior human experience data, and so on. However, there are some important additional requirements and potential dossier gaps to plan for:
- Local Korean-language Documentation: The MFDS requires key trial documents to be provided in Korean (or with Korean summaries). Specifically, the trial protocol, informed consent forms, and subject compensation plan must be submitted in Korean. Even technical sections like the investigational product manufacturing data and nonclinical summaries need at least a Korean synopsis. European sponsors often underestimate the time needed to translate and localize these documents. Ensure your timeline includes translation of the protocol, IB, patient-facing materials, and any investigator training documents into Korean by a qualified team.
- Subject Injury Compensation Plan: Korean regulations mandate that the IND dossier include a plan or insurance policy for compensating trial participants in case of injury. In Europe, sponsors also carry insurance, but MFDS expects formal documentation of this upfront as part of the application. EU sponsors should obtain a Korea-compliant clinical trial insurance certificate or prepare a compensation guideline and have it translated for the IND submission.
- Local Representative Statement: As noted, foreign sponsors must appoint a local authorized representative. The IND dossier will require details of this representative (or local sponsoring entity) and their qualifications. Make sure to include a letter or form confirming the appointment of your Korea rep/CRO to avoid any regulatory hiccup.
- Korean Patient Data Considerations: While not a formal document requirement, MFDS reviewers will examine whether the proposed dose, regimen, and safety data are applicable to Korean participants. European sponsors should be prepared to justify the starting dose or any ethnic sensitivity considerations. If prior trials included Asian populations or if you have pharmacokinetic data bridging to Asians, highlight that. If not, a rationale that the trial population is expected to respond similarly (or any dose adjustments made for Asia) should be provided. This is something EMA applications may not explicitly cover, so it can be a gap for EU-based teams.
- MFDS Format Nuances: Some technical forms (like the application form, electronic submission via the MFDS’s e-submission system) and formatting conventions might differ from EMA. For example, MFDS uses an online Drug Information Portal (NeDrug) for IND submissions. Make sure your local partner is handling the correct format and portal submission. Additionally, Korean INDs require listing all investigator sites and laboratories involved; ensure you’ve identified your Korean sites early to include their details.
European sponsors should also note that IRB approval is separate but simultaneous – MFDS will not permit trial start without IRB clearance from each site. However, you do not need the IRB approvals in hand to submit the IND; these processes can run in parallel (more below). In terms of recent trends, the MFDS in 2025 became a WHO-designated authority (WLA) and overhauled its review teams to further streamline approvals. This pro-innovation stance means that well-prepared applications can sail through quickly. The main pitfalls for foreign sponsors are incomplete dossiers or not meeting local format expectations, which can trigger MFDS queries and slow down approval. By proactively closing the “gaps” – translating documents, appointing a local rep, including an insurance plan, and engaging in a pre-IND meeting – European sponsors can navigate the MFDS IND step with minimal friction.

IRB Review: Realities and Sequencing Challenges
In parallel with MFDS IND approval, sponsors must obtain Ethics Committee approval for the study at each site in Korea. Institutional Review Boards (IRBs) are based at the hospitals or institutions where the trial will be conducted, and each has authority to approve the trial locally. European sponsors may be used to a single central ethics application per country; in Korea, traditionally each site’s IRB reviews the study. This means that if you plan to involve, say, four hospitals, you’ll need four IRB approvals (one per site). The good news is that IRB reviews in Korea are efficient – major hospitals hold IRB meetings 1–2 times per month, and approval is usually obtained ~3 weeks after submission. Recently, a central IRB system has been piloted, where one hospital’s IRB decision can be accepted by others to streamline multi-center studies. However, as of 2026 this central IRB mechanism is not universally adopted, so sponsors should plan assuming separate IRB submissions, unless your sites specifically agree to rely on a central IRB.
The primary sequencing challenge here is timing the IRB approvals relative to the IND. Best practice is to submit to IRBs in parallel with the MFDS IND submission. There is no regulatory requirement to have MFDS approval before an IRB reviews the study; in fact, waiting for sequential approvals will unnecessarily prolong your timeline. Each IRB will evaluate the study’s ethics (informed consent process, risk/benefit, etc.) and often wants to see the same core documents (protocol, IB, consent forms, investigator qualifications). These should be in Korean or bilingual as required by the IRB. Plan for roughly 4–6 weeks for IRB approval at each site – some may be quicker (~3 weeks) if the timing fits their meeting schedule, while others could take a bit longer if revisions are requested. Commonly, an IRB might ask for minor consent form wording changes or clarifications; having a native Korean-speaker on the team (or your CRO) to interface with the IRB is crucial in responding rapidly.
For multi-site trials, synchronization is key. If one site’s IRB approval is delayed (e.g., due to a meeting schedule gap or extra queries), it could become the bottleneck for your First-Patient-In date. One strategy is to identify a lead site – often the site with the most interest or fastest IRB – and use their approval as a central IRB if possible, or at least get that site ready to enroll first. Korea’s recent introduction of mutually recognized IRB review can be leveraged: for example, if Seoul National University Hospital IRB approves a multicenter study, some other hospitals may accept that decision to expedite their sign-off. Sponsors should discuss this possibility during site selection and feasibility. If central review isn’t available for certain sites, ensure that all IRB submissions are made at roughly the same time, soon after IND submission, so that all approvals converge quickly.
Another often overlooked challenge is coordinating contracts and budgets alongside IRB approval. In Korea, the contract with each site (covering investigator compensation, budget, indemnification, etc.) is typically negotiated in parallel with IRB review. European sponsors sometimes wait until after IRB approval to finalize contracts – that is a mistake that can add weeks. Instead, negotiate and even execute site contracts contingent on IRB approval. That way, once the IRB gives the green light, there is no further delay in activating the site. Intoinworld’s step-by-step guide emphasizes treating the IND, IRB, and site initiation processes as an integrated workflow, not sequential steps. By front-loading tasks (translations, contracts, investigator training) during the 4–6 week IND review window, a sponsor can be ready to enroll patients immediately upon regulatory clearance.
In summary, the realities of IRB review in Korea are that you will likely have multiple IRB submissions, but each is fairly quick by global standards (weeks, not months). The main sequencing challenge is ensuring these parallel tracks (MFDS and multiple IRBs) all progress together. European sponsors must adapt from perhaps a single ethics submission mindset to a coordinated multi-IRB strategy. With proper planning – simultaneous submissions, use of any available central IRB option, and parallel contract negotiations – IRB approval in Korea will not be a roadblock but rather a smooth, concurrent step in trial startup.
Safety Reporting & Document Translation: Adapting European SOPs
Operating a Clinical Trial in Korea will require European sponsors to adapt certain standard operating procedures (SOPs), particularly in safety reporting and document management, to comply with local rules. Korea’s pharmacovigilance requirements during Clinical Trials are stringent and must be built into the sponsor’s workflow. All serious adverse events (SAEs) that are unexpected and study-related must be reported immediately to MFDS and the IRB. In practical terms, “immediately” means as soon as possible (often within 24 hours for fatal or life-threatening events). For other serious unexpected adverse drug reactions, Korean regulations typically require notification within 7 days, with a full report within 15 days – analogous to EU’s SUSAR reporting timelines, but sponsors cannot assume that reporting to EudraVigilance or the EMA alone suffices. The MFDS expects direct reports for local trials. European sponsors should update their safety management plan to include MFDS as a reporting recipient for all SUSARs and ensure their local representative/CRO is integrated into safety case processing. Often the local CRO will handle submitting safety reports to MFDS and to all investigators/IRBs in Korea. The key is to clearly assign this responsibility and timing in your SOPs – delays or failure in local SAE reporting can lead to regulatory warnings or even trial suspension.
Document translation is another area where a European sponsor’s usual practices will need adjustment. As mentioned, the MFDS requires core trial documents in Korean, and IRBs require Korean consent forms and often the protocol in Korean. Thus, unlike a trial in many European countries where English documents might be accepted by regulators, in Korea you must budget time and resources for professional translation. This applies not just at the initial submission, but throughout the trial. For example, if the Investigator’s Brochure gets an update or the protocol is amended, the Korean versions must be updated and submitted too. Similarly, patient-facing documents (diaries, questionnaires) should be translated into Korean and culturally adapted as needed for clarity. Back-translations are recommended to ensure accuracy. European SOPs that govern document control should incorporate a step for Korean translation review and approval. Sponsors might establish a translation glossary for technical terms to keep consistency across all documents. Leveraging your local CRO’s bilingual capabilities can streamline this – many Korean CROs have medical writers or translators in-house who are experienced in translating clinical trial documents.
Another SOP adaptation involves data privacy and consent. Korea’s Personal Information Protection Act (PIPA) is robust (similar to GDPR). While consent forms will cover data privacy in Korean, sponsors must ensure their data handling procedures align with Korean law – for instance, how samples or data can be exported out of Korea for analysis. These considerations should be checked with local legal advisors, but generally if you are GDPR-compliant, you’ll meet most PIPA requirements, with the addition that consent forms explicitly mention data transfer overseas if applicable.
In summary, European sponsors must localize their trial operations SOPs for Korea. Safety reporting in Korea requires a rapid local response – make sure your pharmacovigilance team or CRO can notify MFDS/IRBs within days or sooner. All critical documents must be maintained in Korean – set up a workflow for prompt translation of protocols, IBs, case report forms, and patient materials. By planning for these adaptations, sponsors will avoid compliance issues and ensure that the trial in Korea runs according to both global and local standards. Remember that what might be “automatic” in your home country (like informing regulators of SAEs, or providing documents in local language) needs equal attention in Korea. With proper processes, your trial data will be credible and your study compliant in this important market.
Timeline Planning: Best Case vs Worst Case for First-Patient-In
Timeline is everything when sponsors are evaluating Clinical Trial locations. For Korea, one of the biggest draws is the fast startup potential, but realizing that potential requires careful planning. Let’s contrast a best-case scenario versus a worst-case scenario for achieving First Patient In (FPI) – the milestone when the first participant is enrolled in the trial in Korea. This will highlight how strategic planning can save months.
Best-Case Scenario: The sponsor engages in early planning and parallel processing. They hold a pre-IND meeting with MFDS (e.g., 4–6 weeks before official submission) to iron out any issues, then submit a high-quality IND that sails through in ~4 weeks. At the same time (Day 0 of IND submission), they submit IRB applications to all selected sites, leveraging a central IRB for multi-site if available. Within 3 weeks, IRBs grant approvals (perhaps a central IRB covers multiple hospitals in one go). Site contracts, investigator meetings, and drug import licenses are all handled in advance or concurrently. In roughly 6–8 weeks from submission, both MFDS and IRB approvals are in hand. The site activation is immediate and the first patient is screened and dosed shortly thereafter. In an optimal case documented by one sponsor, the clinical trial in Korea was able to start just 5 months after finalizing the protocol – this included time for document prep and all approvals. It’s plausible that for a straightforward Phase I study with a single site, FPI could be achieved in as little as ~8 weeks from regulatory submission. But for a typical multi-center Phase II with more setup steps, ~3–5 months from protocol finalization to first patient is an excellent outcome (and many global sponsors achieve this in Korea).
Worst-Case Scenario: The sponsor treats Korea as an afterthought or follows a sequential approach. Suppose the sponsor finalizes the protocol, then waits to submit the MFDS IND. MFDS takes the full 30 working days (~6 weeks) and issues queries, which take a couple weeks to answer, leading to 8–10 weeks for approval. Only after MFDS approval does the sponsor then start submitting to IRBs at each site. One or two sites miss the nearest IRB meeting deadline, causing a 4–8 week delay for those approvals. Meanwhile, the sponsor also delayed negotiating contracts and translating documents until approvals were in hand. This means after IRB approval, another 4–6 weeks are spent on contract signing, site initiation visits, and training. A competitor trial might have started enrolling patients in the interim, slowing recruitment. In the worst case, this disjointed process can push FPI to 8–9 months after protocol finalization. Indeed, one reported case showed that a sponsor who waited for MFDS approval before preparing IRBs ended up with a startup timeline of 9 months, compared to 5 months for a parallel approach.
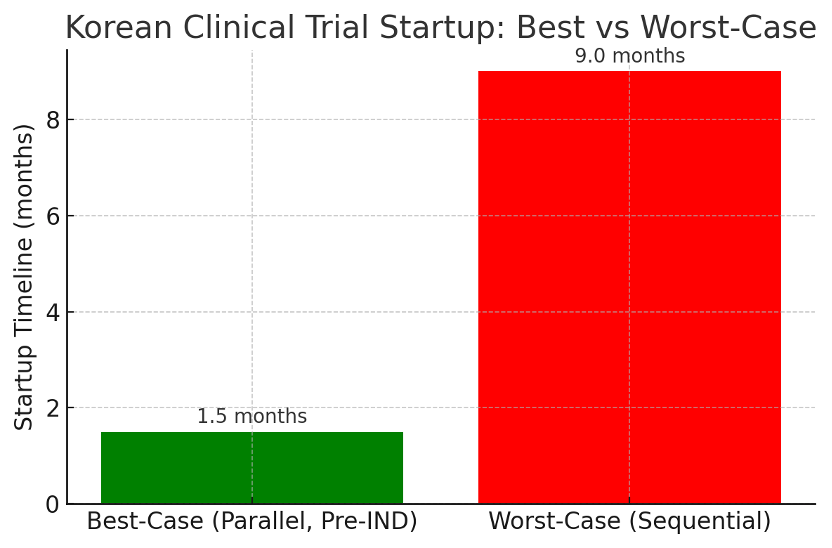
The difference between these scenarios is striking. To achieve the best-case timeline, front-load every feasible activity: obtain agency advice early, prepare all documents in advance, submit everything in parallel, and be ready to initiate sites as soon as approvals land. Korea’s regulatory system allows this parallelism – MFDS and IRBs don’t conflict with each other and are used to running simultaneously. Leverage the central IRB option if possible to cut down multi-site ethics review to a single decision. Also, use tools like the Clinical Research Information Service (CRIS) registration (Korea’s trial registry) in advance, and plan the import permits for the investigational product early. On the other hand, the worst-case teaches that every week of delay at each step compounds. Thus, sponsors should avoid waiting for one step to finish before starting the next. Always ask, “What can I do in parallel?”. In practical terms: don’t wait for MFDS approval to start consenting draft review at sites; don’t wait for IRB approval to draft your site budgets, etc.
In conclusion, Korea offers a best-case startup timeline that is among the fastest in the world for Clinical Trials, but sponsors only reap this benefit if they execute a parallel, well-coordinated startup plan. By contrast, if a team brings a slow, linear mindset, Korea’s advantage can be underutilized. European sponsors planning trials for 2026 should aim to replicate the best-case scenario through diligent planning – doing so can mean reaching critical trial milestones (like FPI) months earlier, which in drug development can translate to significant competitive and financial gains.
Strategic Checklist for Sponsors Planning Korea Clinical Trials (2026)
To help European sponsors ensure nothing is overlooked, below is a strategic checklist that can be copied and used during planning. This checklist highlights the essential steps and best practices discussed above:
- Appoint Local Representative Early: Designate a Korea-based entity or CRO to fulfill local sponsor duties and MFDS liaison requirements. Ensure contracts with the local representative/CRO clearly outline responsibilities (regulatory submissions, safety reporting, etc.).
- Prepare Korea-Ready Documents: Compile all required IND dossier materials (protocol, IB, CMC data, etc.) and translate key documents into Korean. This includes the protocol synopsis, informed consent forms, patient-facing materials, and compensation plan.
- Pre-IND Consultation with MFDS: Schedule an MFDS meeting before IND submission to validate your strategy and address any Korea-specific questions. Incorporate MFDS feedback to avoid IND review delays.
- Parallel MFDS & IRB Submissions: Submit the IND application and IRB applications concurrently. Do not wait for MFDS approval to initiate IRB reviews at sites. Aim to have IRB approvals progressing during the 30-day MFDS review window.
- Leverage Central IRB (if available): If conducting a multi-center trial, use a central IRB review process or agree on IRB reciprocity among sites to speed up ethics approval. If not, ensure all site IRB submissions are synchronized for simultaneous approvals.
- Integrate Safety Reporting Locally: Update pharmacovigilance SOPs to report SAEs promptly to MFDS and local IRBs. Define whether the sponsor or local CRO is responsible for reporting; ensure 24/7 readiness to meet Korea’s reporting timelines.
- Conduct Parallel Site Initiation Tasks: While awaiting approvals, negotiate site contracts, complete budget agreements, import licenses, and translations of study tools. Train investigators on protocol and Korea-specific GCP nuances in advance.
- Plan Timeline with Buffer: Although Korea is fast, build in a buffer for unexpected MFDS queries or IRB comments. Identify “critical path” items (e.g. drug import release) and have contingency plans to prevent these from delaying first-patient-in.
- Validate GCP Compliance & Data Plans: Ensure your trial monitoring, data capture, and quality management plans meet Korean (KGCP) and ICH standards. Consider a dummy FDA/EMA compliance check on the protocol – since Korean data can be used for EU approvals, design the trial to global standards.
- Engage a Local CRO Partner: If not already done, engage a reputable Korean CRO to navigate cultural, linguistic, and regulatory nuances. Their insight can prevent common pitfalls (e.g. document formatting issues, missing local lab accreditations, etc.) and keep the trial on track.

Next Steps: Request a Quote & Stay Informed
With the 2026 landscape offering rapid approvals and high-quality Clinical Trials in Korea, European sponsors can seize this opportunity by executing a well-planned strategy. Intoinworld is here to support your journey – from regulatory consulting to full-service CRO execution in Korea. Ready to take the next step? Request a Quote for your upcoming trial to receive a tailored plan and timeline. Our team will provide a detailed proposal and budget, helping you evaluate Korea’s fit for your project.
Stay ahead on evolving Korea clinical trial requirements by subscribing to Intoinworld’s updates. Join our Newsletter (visit our Newsletter page) for the latest regulatory news, trial trends, and case studies delivered to your inbox. By staying informed and partnering with experienced local experts, European pharma and biotech companies can confidently expand their Clinical Trial programs into Korea – leveraging its speed, quality, and innovation for global success.
FAQ: Clinical Trials in Korea – European Sponsors’ Common Questions
Q1. How long does the Clinical Trial approval process take in Korea (MFDS IND & IRB)?
A1. Korea offers a very fast startup. MFDS review of an IND application is officially 30 working days, but in practice European sponsors see approval in ~4–6 weeks. Crucially, IRB ethical reviews at hospitals are done in parallel and take around 3 weeks on average per site. By submitting to MFDS and IRBs simultaneously, the total preparation time until trial initiation is typically only 6–8 weeks. This is much faster than in many other countries (e.g. Japan or China), making Korea’s startup timeline one of the most attractive globally. With perfect planning (and use of MFDS pre-consultation), some trials have even started in as little as 4–5 weeks after submission, whereas a poorly planned sequence could take a few months.
Q2. Do European sponsors need a local entity or partner to conduct Clinical Trials in Korea?
A2. Yes – foreign sponsors must have a local presence via either a legal entity in Korea or a designated local representative. The MFDS considers overseas sponsors without a local office as not meeting all obligations. In practice, most European companies appoint a Korean CRO as their legal representative to handle regulatory interactions. Alternatively, if a sponsor has a Korean subsidiary, that entity can act as the trial sponsor. Engaging a local partner is highly recommended because they understand Korean regulations and language. While not an absolute requirement to use a CRO (if you have your own entity), working with an experienced Korea CRO greatly smooths the process– they prepare submissions, interface with MFDS/IRBs, and manage sites on the sponsor’s behalf. This ensures compliance and efficiency for European sponsors navigating Korea’s system.
Q3. Are data from Korean Clinical Trials accepted by EMA and FDA for approvals?
A3. Yes. Clinical data from trials in South Korea are accepted by both the European Medicines Agency (EMA) and U.S. FDA, provided the trials are conducted under ICH-GCP standards. Korea is an ICH member, and its regulatory authority (MFDS) is a WHO-Listed Authority, so its trial oversight is globally recognized. In fact, many global drug applications include Korean trial data – for example, a Korean Phase II oncology study can be used in an EMA submission if it’s well-designed. Both EMA and FDA have provisions (21 CFR 312.120 for FDA) that allow foreign trial data to support approvals as long as audits can verify data integrity and ethical conduct. Sponsors should ensure the Korean trial protocol aligns with international expectations (endpoints, patient population, etc.). With that, Korean trial results can definitely form part of a European dossier or FDA filing. This means European sponsors can run a trial in Korea and use the results globally, avoiding the need to duplicate studies.
Q4. What documents are required for a Clinical Trial application (IND) to MFDS?
A4. The MFDS IND application dossier is similar to a European CTA file, with some added specifics. Key documents include: the clinical trial protocol, Investigator’s Brochure, information on investigational product quality and manufacturing (CMC data with GMP compliance proof), non-clinical study results, any available clinical data or previous human experience, and details on the trial sites and investigators. Additionally, MFDS requires a document on compensation for trial subjects (insurance policy or compensation plan), which should be prepared in case of trial-related injuries. All submissions must be in the CTD format and importantly, several documents need Korean translations or summaries: the protocol (or at least its summary) in Korean, the informed consent form in Korean, and a Korean summary of nonclinical and clinical data. In short, European sponsors should be ready to submit a full technical dossier akin to an IMPD, plus ensure patient-facing documents and certain summaries are in the Korean language for the MFDS review.
Q5. How do Korea’s safety reporting requirements differ from Europe’s during a Clinical Trial?
A5. Korea’s safety reporting requirements are stringent and in some ways more hands-on locally. In Europe, sponsors report SUSARs to EudraVigilance; in Korea, any serious and unexpected adverse event related to the study drug must be reported promptly to the MFDS and to all participating investigators/IRBs. “Promptly” is generally interpreted as immediately or within 7 days for very serious cases (with a follow-up in 15 days), similar to EU timelines but requiring direct local notification. The sponsor (or their local representative) should submit safety reports in Korean through the MFDS’s system. It’s not enough to just inform EMA/FDA; Korea must be informed separately. European sponsors need to integrate their pharmacovigilance workflow so that as soon as a SUSAR is identified, a report is filed with MFDS (in addition to global regulatory reporting). Another difference: Korea often requires an annual safety report to MFDS on the trial’s safety status (akin to a DSUR in Europe). Sponsors should also ensure the informed consent form in Korea mentions that safety information will be collected and reported to authorities. In summary, while the types of events to report are the same as Europe, the reporting route is local – having a local PV contact or CRO is essential to meet the Korean timeline and format requirements.




