1. Background and goals
The amended Advanced Regenerative Bio Act, effective 21 February 2025, marks a policy turnaround by expanding access to advanced regenerative medicine and strengthening patient‑safety management. The law lifts research‑subject restrictions so that therapies can be provided not only through clinical trials but also as routine treatment. It also enhances transparency in pricing and monitoring of adverse reactions.
2. Summary of major changes
Removal of subject restrictions and introduction of treatment: Whereas the previous law focused on severe and rare diseases, the amendment allows patients with mild or common conditions to receive regenerative treatments. The concept of advanced regenerative‑medicine treatment enables institutions to offer therapies outside clinical trials, provided they obtain approval from an ethics committee and meet safety standards.
Cost disclosure and patient rights: Sponsors must include purpose, target population, safety/efficacy evidence and cost calculations in the treatment plan. The Ministry of Health is required to publish annual treatment costs, and patients must be informed of expected out‑of‑pocket expenses. Informed‑consent procedures now include data protection and contingencies for treatment discontinuation.
Enhanced oversight: Ethics committees can request progress data for both clinical trials and treatments, and the health minister has new authority to conduct site inspections. Subordinate regulations clarify adverse‑reaction definitions, broaden the scope of safety investigations and expand expert advisory panels.
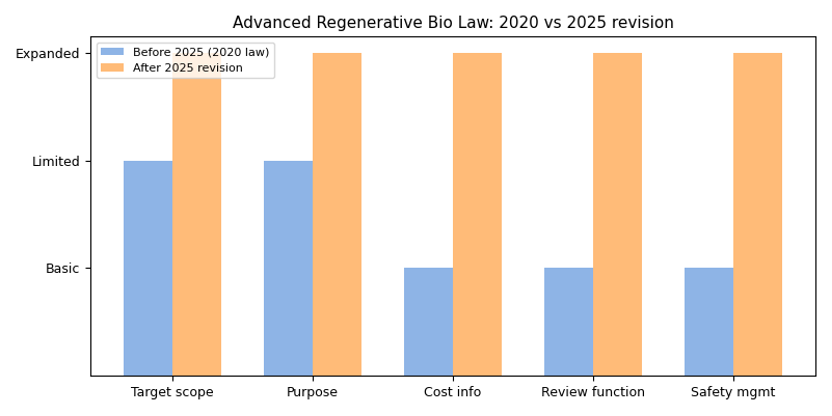
Comparison diagram: The following graphic compares the 2020 law with the 2025 amendments, showing expansions in target scope, purpose, cost information, review functions and safety management.
3. Practical guidance by stakeholder
Sponsors/developers: From the design stage, sponsors should consider treatment applications for a broad range of conditions, prepare documentation that includes safety/efficacy evidence and cost estimates, and verify that partnering institutions are designated regenerative‑medicine providers.
CROs: Contract research organizations must strengthen their document‑preparation capabilities, including templates for treatment plans, informed‑consent forms and cost explanations. They should establish processes for submitting documents and responding to monitoring visits, and build systems for recording adverse events, reporting costs and tracking patient follow‑up.
Medical institutions/researchers: Institutions should ensure they meet the designation requirements for regenerative‑medicine providers and train staff accordingly. They must understand the steps for ethics‑committee review, cost disclosure and adverse‑reaction monitoring and prepare documentation and internal checks to handle site inspections.
4. Insights from Korea’s clinical‑trial environment
Korea ranks sixth globally for sponsor‑initiated clinical trials; Seoul was the world’s top city for trial activity between 2017 and 2023. These statistics underline the international trustworthiness of Korean data.
Recognition by regulators: Korea is an ICH member, and all trials adhere to ICH‑GCP. The FDA and EMA officially accept overseas data that meet these standards. Korean participation in multinational trials—such as Celltrion’s CT‑P13 biosimilar approved by the EMA—demonstrates the value of Korea‑generated data.
Approval timeline: Clinical‑trial protocols must receive MFDS IND approval and ethics‑committee review. IND review typically takes 4–6 weeks, and IRB review around 3 weeks; a central IRB system expedites multicentre studies. The following timeline summarizes the steps.

Infrastructure and data management: Approximately 200 institutions are certified to conduct trials, many of them large tertiary hospitals. Korea’s universal healthcare system and electronic medical records enable rapid recruitment—about 25 % faster than the Asian average. Data are managed through electronic data‑capture systems linked to hospital records, reducing errors and ensuring compliance with ICH E6(R2).
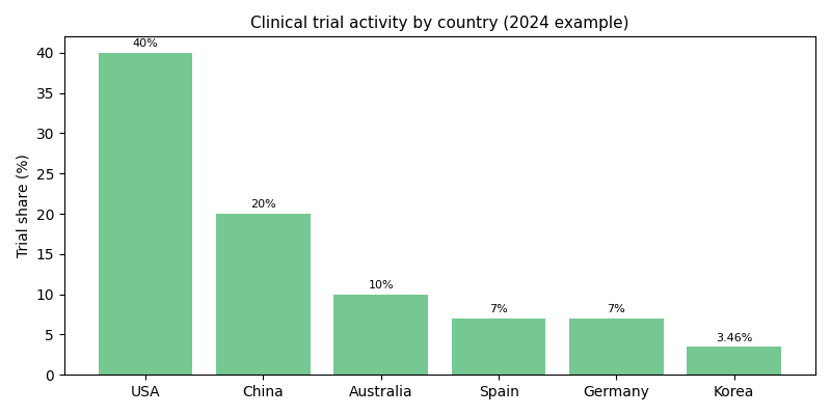
Global share: The following bar chart illustrates the share of clinical‑trial activity by country in 2024. While the USA and China dominate, Korea holds about 3.46 %, placing sixth.
5. Conclusion and how Intoinworld can help
The February 2025 amendments shift Korea’s regulatory framework from a research‑centric model to one that prioritizes patient access and safety. Start‑ups and developers need to integrate treatment planning into early trial design, CROs must bolster documentation and monitoring capabilities, and medical institutions should prepare for designation and site inspections. Korea’s robust infrastructure and strict adherence to ICH‑GCP provide a strong foundation for global regulatory strategies.
Intoinworld understands these regulatory changes and offers integrated support for drafting treatment plans, cost‑disclosure systems, site‑inspection readiness and ethics‑committee strategies. For more details on Korea’s clinical‑trial environment, visit our clinical trial information page. We invite you to request a quotation or subscribe to our newsletter to stay informed about the latest developments.
Frequently asked questions (FAQ)
Q1. Who benefits from the amendments to the Advanced Regenerative Bio Act?
A1. Patients with mild or common conditions now qualify for regenerative‑medicine treatment as subject restrictions have been removed. Treatments must be conducted at designated regenerative‑medicine institutions under ethics‑committee approval.
Q2. Are Korean clinical‑trial data accepted by the FDA and EMA?
A2. Yes. Because Korea adheres to ICH‑GCP, the FDA and EMA officially accept Korean data that meet ethical and scientific standards. Supplemental studies may be required depending on the regulatory agency’s specific demands.
Q3. What documentation do companies need to comply with the amended law?
A3. Sponsors and CROs must prepare treatment plans that include purpose, target population, safety/efficacy evidence and cost calculations, along with informed‑consent forms, cost‑explanation materials and adverse‑reaction monitoring plans.

