Introduction: A Growing Cancer Burden Meets Evolving Clinical Trials Landscape
Korea’s projected cancer burden remains large – and it’s growing. In 2025, national estimates foresaw 304,754 new cancer cases and 84,019 cancer deaths. By extrapolating recent trends, 2026 is expected to bring an even higher incidence of cancer cases (around 317,000) with a relatively stable number of deaths (mid-84,000s). This increase in incidence underscores a critical point for Clinical Trials planners: a bigger pool of cancer patients does not automatically translate to an equally large pool of trial participants. Instead, Korea’s 2026 oncology landscape will challenge global pharmaceutical and biotech sponsors to think beyond sheer numbers – focusing on patient mix, eligibility filters, and operational realities in clinical trials.
This article provides a data-backed outlook on Korea’s cancer Clinical Trials environment for 2026. We distinguish between raw cancer incidence and the recruitable patient pool for trials, examine 2025→2026 projected shifts in the oncology population, and highlight factors like aging patients, prior treatment burden, and screening impacts on trial enrollment. We also discuss how stage distribution changes and evolving standard-of-care in Korea create new opportunities and constraints for oncology Clinical Trials. Throughout, we’ll draw planning implications to help sponsors design smarter eligibility criteria, endpoints, and monitoring plans for their Clinical Trials in Korea’s oncology sector.
(Read on for analysis of national cancer data and practical tips. For additional context on Korea’s overall trial infrastructure and regulatory advantages, see Korea’s strategic clinical trial hub status after MFDS reforms.)
2025 Data Baseline: High Incidence, Concentrated in Key Cancers
Before forecasting 2026, it’s important to anchor on the latest national data. Korea’s 2025 cancer projections estimated 304,754 new cases (all cancers combined). Notably, just six cancer types – thyroid, colorectal, lung, breast, prostate, and stomach – account for about 63.8% of those new cases. Figure: Top 6 Projected Cancer Incidences in Korea (2025) below illustrates how these cancers dominate the incidence pool.

2025 Data Baseline: High Incidence, Concentrated in Key Cancers
Before forecasting 2026, it’s important to anchor on the latest national data. Korea’s 2025 cancer projections estimated 304,754 new cases (all cancers combined). Notably, just six cancer types – thyroid, colorectal, lung, breast, prostate, and stomach – account for about 63.8% of those new cases. Figure: Top 6 Projected Cancer Incidences in Korea (2025) below illustrates how these cancers dominate the incidence pool.
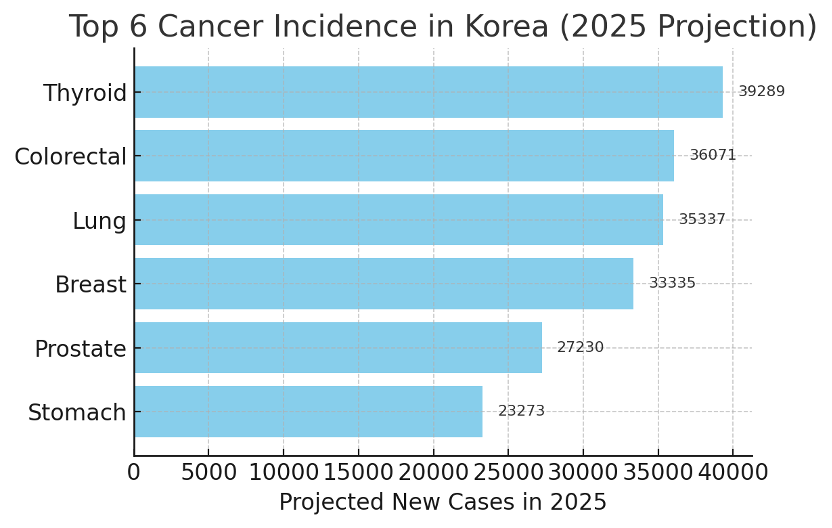
Projected top six cancers by new cases in Korea (2025). Thyroid cancer leads with ~39k cases, followed by colorectal and lung (each ~35k), breast (~33k), prostate (~27k), and stomach (~23k).
For trial sponsors, this concentration has a crucial implication: “Top incidence” does not equal “top recruitability.” For example, thyroid cancer – the single most common cancer in Korea by incidence – often isn’t a priority for global Phase 1–3 drug development, as its standard treatments are effective and many cases are early-stage. In contrast, cancers like lung, colorectal, and breast (also very common in Korea) align with numerous international Clinical Trials and novel therapeutics pipelines. These cancers tend to have multiple lines of therapy, unmet needs in advanced stages, and biomarker-defined subpopulations, making them fertile ground for oncology clinical trials. Therefore, sponsors evaluating Clinical Trials in Korea should map incidence data to trial-relevant patient pools, focusing on where the recruitable patients truly are. In practice, that means prioritizing indications where large patient numbers intersect with trial eligibility – e.g. lung, breast, colorectal, gastric, and other tumors where investigational drugs target specific mutations or resistant disease. It also means recognizing when a high-incidence cancer (like thyroid) might yield relatively few candidates for a given clinical trial (due to early-stage surgery cures or lack of need for experimental therapy).
From 2025 to 2026: More Cases, More Survivors – and a Smaller Late-Stage Pool?
Using 2025 as a springboard, what can sponsors expect in 2026? The government’s official 2026 cancer forecast isn’t published yet, but we can build a practical planning estimate. The year-on-year increase from 2024 to 2025 was +12,533 new cases (from 292,221 to 304,754). If we apply a similar delta, 2026 could see roughly 317,000 new cancer cases nationwide. In contrast, annual cancer deaths changed very little (only +249 from 2024 to 2025). So while incidence keeps rising, mortality is nearly flat – a trend likely to continue into 2026.
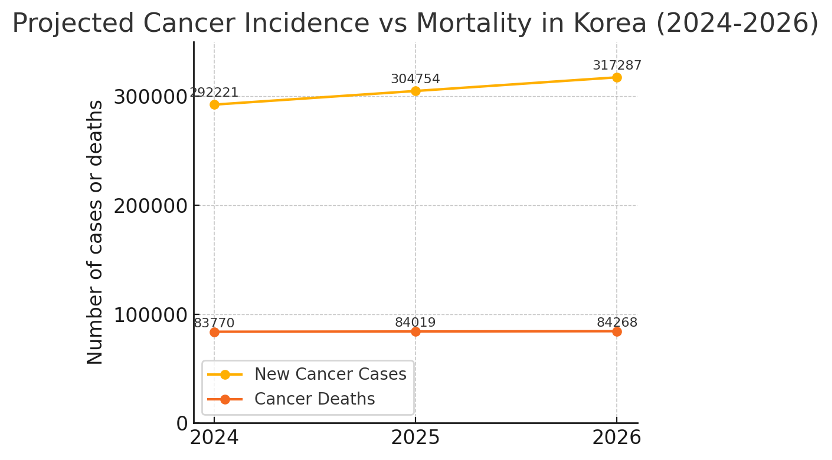
Projected cancer incidence vs mortality in Korea (2024–2026). Annual new cases (blue line) are rising significantly, whereas annual cancer deaths (orange line) show only a slight increase. This divergence suggests improving survival and earlier detection.
Why do incidence and deaths move differently? Rising incidence paired with slower mortality growth points to improvements in outcomes. Thanks to early detection and better treatments, more patients are surviving longer after diagnosis. Korea’s national data confirms this: the 5-year relative survival rate for all cancers diagnosed 2018–2022 reached 72.9%, with over 2.59 million people alive who had been diagnosed with cancer (prevalent cases as of 2022). In short, Korea has more people living with cancer than ever before, not just more new cases.
For trial planning, this survivorship-heavy reality means many 2026 trial candidates will have long treatment histories. More patients will be multiple years post-diagnosis or on second, third-line therapies rather than naïve to treatment. The high survival and prevalence also indicate earlier-stage diagnoses are more common now (since many live beyond five years). Sponsors should anticipate that a typical Korean patient in 2026 may be older and previously treated, rather than an idealized “clean slate” patient. Eligibility criteria in Clinical Trials must accommodate prior lines of therapy and comorbidities, or risk excluding much of the real patient population.
Incidence vs. Recruitable Patients: Bridging the Gap
It’s worth reiterating for 2026 planning: having X new cancer cases in Korea does not equal X patients available for your trial. There’s a drop-off at each step from incidence to consented enrollment. Consider all the filters a patient must pass through to be “trial-eligible”:
- Stage and line of therapy: Among new cases, only a subset will be at the stage or line your clinical trial requires (e.g. metastatic second-line). Earlier-stage patients or those already cured by surgery won’t qualify for many drug trials.
- Standard-of-care (SOC) received: By 2026, many patients will receive effective SOC therapies before a trial is an option. If a new therapy (like an adjuvant immunotherapy) becomes standard, fewer patients remain in the late-line setting for trials. Sponsors must track how SOC shifts year to year in Korea.
- Biomarker availability: Incidence counts often ignore biomarker status. For targeted therapy trials, only the fraction of patients with the requisite mutation or protein expression are candidates. For example, among lung cancer cases, maybe only 15% have EGFR mutations – and only those can join an EGFR inhibitor trial.
- Health and performance status: Elderly patients or those with comorbidities might not pass stringent inclusion criteria, even if they technically have the cancer of interest. By 2026, with an aging population, more patients will have lower performance status (PS) or other illnesses that cause screen failures in Clinical Trials.
- Competing trials: Korea is a busy research hub; patients often have multiple trial options. A large incidence cancer like hepatocellular carcinoma might have dozens of concurrent trials competing for the same pool of patients, effectively reducing availability for any single clinical trial.
In summary, the funnel from incidence to recruitable pool narrows considerably. Sponsors planning Clinical Trials in Korea for 2026 must explicitly model this funnel, rather than relying on raw patient numbers. As one Korea oncology specialist noted, “Korea has X cases” is a weak statement unless you translate it into recruitable pool logic. The most successful trial projections will quantify how many patients are likely to meet all criteria and actually enroll, after factoring in stage distribution, prior treatments, biomarkers, PS, and trial competition.
The Aging Patient Profile: Implications for Clinical Trials in 2026
One of the most profound shifts in Korea’s cancer demographics is aging. Korea is an aging society overall, and cancer patients are no exception – in fact, improved survival means the average oncology patient is older. This is not an abstract trend; it’s measurable in the clinic. For example, a nationwide survey of gastric cancer patients found the share of patients aged ≥71 jumped from 9.1% in 1995 to 31.7% in 2023. Within a few decades, seniors have gone from a small minority to nearly one-third of that cancer’s patient population. (See Figure: Aging of Gastric Cancer Patients below.)
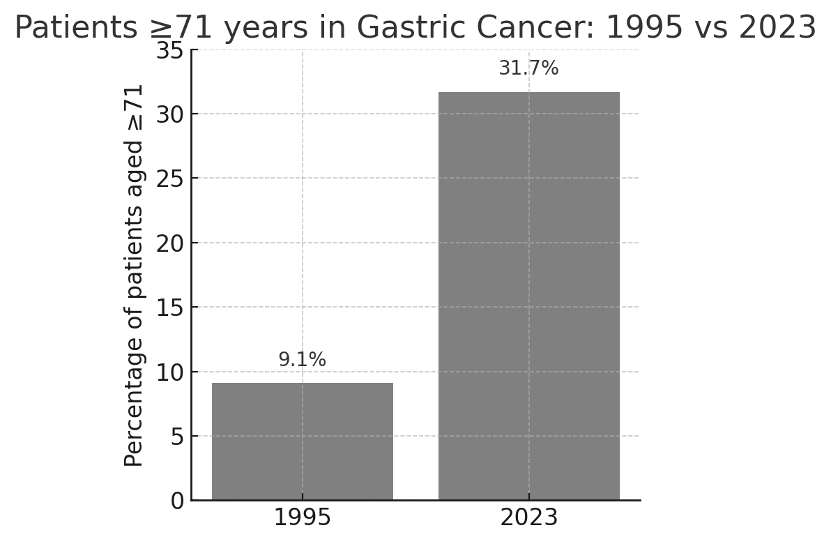
Aging of cancer patients in Korea: In gastric cancer, only ~9% of patients were ≥71 years old in 1995, but by 2023 that share rose to ~32%. Similar trends of older patient populations are observed across many tumor types.
By 2026, we can expect the proportion of elderly cancer patients to be even higher. Why does this matter for Clinical Trials? Older patients often bring multiple challenges:
- Comorbidities and medications: An older trial candidate is more likely to have other chronic diseases (diabetes, heart disease, etc.) and be on concomitant medications. This can complicate trial protocols (drug interactions, exclusion criteria) and increase the risk of adverse events.
- Strict eligibility criteria conflicts: Many trials historically excluded patients based on factors common in older adults (e.g. impaired organ function, prior cancers, frailty). If inclusion/exclusion criteria aren’t updated to be geriatric-friendly, a growing segment of real patients will be deemed ineligible.
- Safety and monitoring: Toxicities may be more frequent or severe in older patients, and they might require closer safety monitoring. Site staff will need to watch for, for instance, higher risk of neutropenia or cardiac side effects in older cohorts during Clinical Trials.
- Logistics and retention: Elderly patients may have mobility issues or difficulty traveling frequently to hospitals. Trials with heavy visit schedules or onerous procedures risk drop-out or non-compliance in this group, affecting data quality.
In planning Korea trials for 2026, sponsors should calibrate protocols to an older patient profile. This might involve broadening eligibility (if medically justifiable) to include well-managed comorbid conditions, simplifying visit schedules, and investing in patient support (e.g. travel assistance or home health visits). Clinical Trials that assume a “young, fit” participant pool may struggle to enroll in Korea now; screen failure rates will spike when the actual median patient is 70 and on multiple medications. Proactively adjust your trial design – endpoints, dosing, monitoring frequency – to be feasible and safe for the real-world patient. As Korea’s data shows, aging is an inevitable part of the oncology clinical trial landscape here.
Early Detection and Evolving Therapy: Stage Shift in Korean Oncology
Another dynamic trend in Korea is the shift toward earlier-stage disease at diagnosis for several major cancers. Decades of cancer screening efforts (and public awareness) are paying off: the proportion of cancers caught at a localized (early) stage has risen significantly. For instance, in stomach cancer – where Korea has an intensive national screening program – early gastric cancers accounted for 63.1% of cases in 2023. That means the majority of newly diagnosed gastric cancer patients can undergo curative surgery or endoscopic resection, rather than presenting with incurable advanced disease.
This “stage migration” affects the Clinical Trials landscape:
- More early-stage patients create opportunities for trials in adjuvant, neoadjuvant, or minimal residual disease (MRD) settings. When many patients are diagnosed early, there’s a larger pool for studies aiming to prevent recurrence or eliminate microscopic disease post-surgery. Trials of novel therapies given before or after surgery (e.g. immunotherapy or targeted drugs as adjuncts) become more feasible and important.
- Fewer late-stage patients in some cancers can tighten the recruitment pool for metastatic or refractory disease trials. Using gastric cancer as an example: if only ~37% of new cases are advanced at diagnosis, and among those many respond to first-line treatment, the pool for a third-line gastric cancer trial is a fraction of total incidence. Sponsors in areas like breast, colorectal, or gastric cancer must account for improved standard-of-care outcomes that reduce the available patients for later-line Clinical Trials. In contrast, cancers without effective early detection (e.g. pancreatic, certain lung cancers) may not see this effect as strongly – those remain largely advanced at diagnosis.
- Evolving standard-of-care: Stage shift often accompanies new treatment standards. For example, widespread use of adjuvant immunotherapy in some cancers could become SOC by 2026, raising the bar for trial comparators and endpoints. A trial design from 2018 might be obsolete in 2026 if it assumed patients hadn’t received a now-standard therapy. Global sponsors must stay current on Korea’s treatment landscape, which can change year by year.
In practical terms, trial sponsors should map the stage distribution and treatment algorithm in Korea for each indication they are considering. If early detection programs (like LDCT lung cancer screening, mammography, colonoscopy, etc.) are expanding the early-stage population, consider pivoting some Clinical Trials to those earlier settings where patients are more plentiful. Conversely, if planning a late-line Clinical Trial, double-check how many patients will realistically still need that option after receiving modern first- or second-line therapies. Flexibility in trial design – e.g. including multiple lines of therapy or stratifying by prior treatment – can help adapt to this shifting landscape.
(For a deep dive into how new biomarkers and treatment strategies are changing oncology trials, see 2026 ADC Clinical Trial Trends: From HER2-Low to TROP-2 and Korea’s Opportunity, which discusses how expanding biomarker-defined populations are altering trial designs.)
Planning Implications for Global Sponsors Running Clinical Trials in Korea
What do these trends mean for a global pharma/biotech team evaluating Korea as a Clinical Trials site in 2026? In essence, sponsors should convert these macro trends into concrete operational decisions. Below is a practical planning checklist to leverage Korea’s data for trial success:
- Build an indication-specific patient map, not just a country-level slide. Start with Korea’s 2025 incidence and mortality data for your cancer of interest. Break it down by stage, line of therapy, and major subtypes. Validate how patients flow through the local system (e.g. what % get surgery, what % get first-line targeted therapy). This map should highlight where patients drop out of the pool (due to cure, SOC, etc.) and where recruitable patients accumulate. For example, if colorectal cancer has 36k cases a year, find out roughly how many reach second-line metastatic – that’s your target pool for a clinical trial in a new second-line drug.
- Account for aging and comorbidities in eligibility. Proactively identify criteria in your protocol that might exclude older or sicker patients, and assess if they are truly necessary. Could a slightly lower renal function or a well-controlled comorbidity be allowed without compromising safety? By adjusting these, you include a larger share of Korea’s real 2026 patient cohort. Also plan for potentially higher screen fail rates if your trial demands very fit patients – sites in Korea can then over-enroll on screening to compensate.
- Apply “real-pool filters” to incidence when forecasting enrollment. When setting recruitment targets, explicitly factor in filters such as biomarker positivity rates, prior line exclusions (e.g. no prior IO therapy – what % of patients will that rule out?), and competing trial density in Korea. If, say, 30% of lung cancer patients have EGFR mutations and your trial needs EGFR+, multiply incidence by 0.3. If 50% of those might have received a new standard drug already (making them ineligible), multiply again by 0.5. Do this cascade of adjustments to get a realistic pool size. This protects you from overly optimistic assumptions and helps right-size your Clinical Trials recruitment goals.
- Design with the current standard-of-care and stage mix in mind. Ensure your trial’s control arm and inclusion criteria reflect Korea’s 2026 standard treatments. If early-stage diagnoses are common, perhaps consider adding an adjuvant cohort or designing the trial to also enroll high-risk stage II patients, etc. Make endpoints relevant to an era of longer survival (e.g. disease-free survival if overall survival will be prolonged by subsequent therapies). Basically, localize your trial design to Korea’s oncology trends so that it fits into patients’ treatment journey.
- Plan operational resources for a complex patient profile. Higher prevalence of cancer survivors means more protocol deviations if not carefully managed – patients may join Clinical Trials with residual toxicities or lab abnormalities from prior treatments. You may need more intensive site monitoring and data cleaning. Consider increasing monitoring frequency or including more on-site CRA visits, especially early in the trial, to catch safety or data issues. Training site staff on how to manage older patients or those on polypharmacy in the trial context can also improve data quality and patient safety. Essentially, use the knowledge of “who the 2026 patient is” to inform everything from trial supplies (e.g. need for larger print consent forms for older eyes?) to DSMB oversight focus.
By implementing these strategies, sponsors can make the most of Korea’s strengths – a large patient population and high research quality – while mitigating risks posed by demographic and healthcare trends.
Frequently Asked Questions (FAQs)
Q: Why does a large cancer incidence in Korea not equal a large pool of trial patients?
A: Incidence is the total number of new cancer cases, but Clinical Trials can only recruit patients who meet strict criteria. Many patients are filtered out due to early stage (cured by standard treatments), wrong line of therapy, no required biomarker, poor health, or other factors. For example, out of thousands of new cases, only a fraction might have advanced disease and the biomarkers a trial needs. Thus, clinical trial planners must adjust incidence figures to estimate a realistic recruitable pool.
Q: How is Korea’s aging population affecting oncology Clinical Trials?
A: Korea’s cancer patients are getting older on average. An older population means trial participants with more comorbid conditions and prior medications. This trend leads to higher screen failure rates if trials have overly strict health criteria. It also raises the risk of side effects and drop-outs. Sponsors need to design Clinical Trials protocols that accommodate older patients – for instance, by relaxing certain exclusions, simplifying visit schedules, and enhancing safety monitoring for geriatric participants.
Q: What impact does early detection have on Clinical Trials in oncology?
A: Earlier detection of cancers (through screening programs) means more patients are diagnosed at curable stages. While that’s good for patients, it can shrink the pool of late-stage patients available for trials of new drugs. On the other hand, it opens up new trial opportunities in adjuvant or neoadjuvant settings (testing treatments around the time of surgery) and in preventing recurrence (MRD trials). Sponsors must adapt by focusing Clinical Trials on the disease stages where patients are now more commonly found in Korea. If a cancer’s late-stage incidence is dropping due to screening, trials targeting only end-stage patients may struggle to enroll.
Q: What are the key 2026 oncology trends in Korea that global sponsors should consider?
A: Key trends include rising incidence (more new cases each year), higher survival (more people living with cancer), an aging patient demographic, and shifts in stage at diagnosis. There’s also rapid uptake of new standard treatments. For sponsors, this means a larger but older and more pre-treated trial population. Trials need to accommodate prior lines of therapy and comorbidities. Endpoints may need to capture longer survival times. And because standard-of-care is a moving target, trial designs must be kept up-to-date. Essentially, Korea’s 2026 Clinical Trials environment demands flexibility and local insight in planning.
Q: How can sponsors improve their clinical trial success in Korea’s oncology sector?
A: Sponsors should leverage data in planning. First, use Korea’s national cancer statistics to identify where the recruitable patients are (by cancer subtype, stage, etc.). Second, tailor inclusion criteria to real-world patient profiles in Korea, which in 2026 means older patients with prior treatments. Third, align your trial with the local standard-of-care – e.g. choose control regimens and endpoints that make sense given what Korean patients typically receive. Engaging local Key Opinion Leaders (KOLs) or a local Clinical Research Organization (CRO) can provide on-the-ground insights. Finally, plan for robust site support: ensure your monitoring, data management, and enrollment strategies reflect the complexity of patients and the competitive Clinical Trials landscape in Korea. By doing so, sponsors can tap into Korea’s advantages (high patient volume, excellent medical infrastructure) and mitigate risks, leading to more efficient trials.
Korea’s cancer Clinical Trials environment in 2026 will be characterized by abundant patient numbers set against the backdrop of demographic shifts and high standards of care. Global pharma and biotech sponsors can greatly benefit from including Korea in their trial portfolios – but success requires going beyond surface statistics. By understanding that cancer incidence ≠ trial eligibility, anticipating the needs of older and previously treated patients, and adjusting to a landscape of earlier diagnoses, sponsors can design trials that enroll on time and generate high-quality data. Korea offers a strategic mix of a large patient pool, world-class investigators, and regulatory acceptance of data globally. To capitalize on this, sponsors must plan diligently, staying data-driven and patient-centric.
If you are interested in launching or expanding your Clinical Trials in Korea, our team can help. Click the Quotation Request button below to get a detailed proposal on how we support trial setup, patient recruitment, and regulatory navigation in Korea. Additionally, subscribe to our newsletter for monthly insights on Korea’s latest clinical trial trends (so you never miss an update on key changes like MFDS policies or oncology developments). Let us be your bridge to successful Clinical Trials in Korea’s dynamic oncology landscape.