When you plan a clinical trial in Korea, the first thing you need is a clear view of “how many weeks this will realistically take.” A start-up timeline is not just the MFDS review window. It is the full start-up clock—from feasibility and IND package preparation to IRB submissions, contracts and budgets, vendor and system setup, site activation, and finally first patient in (FPI). Korea can move quickly when parallel workstreams are designed well. But if operational items slip—contracts, translations, vendors, system validation, drug supply—the overall timeline can shift easily. This guide summarizes practical time ranges by phase, the points where projects most often stall, and the variables that move both timeline and budget.
At a glance: typical total duration
Exact timing depends on protocol complexity and readiness. In practice, global teams often plan with three planning bands.
Fast scenario: about 8–10 weeks
Assumptions: a complete submission package, rapid contract/budget alignment, limited vendor count, and no drug supply constraints.
Typical scenario: about 12–16 weeks
Most new global studies fall in this range.
Higher delay risk: 16–24+ weeks
Common drivers: prolonged contracting, repeated translation revisions, slower vendor/system setup, central imaging/adjudication requirements, import/labeling constraints for investigational product, or feasibility assumptions that are too optimistic.
Use these ranges as a baseline for internal approvals and risk management. The sections below explain what extends timelines and how to plan around it.
Korea start-up roadmap
Most Korea start-ups follow a consistent flow:
Strategy alignment (define Korea’s role and success criteria) → feasibility and site strategy → IND preparation and submission → IRB/site submissions and approvals → contracts and budget finalization → vendor and system setup (data capture, randomization and drug supply systems, electronic outcome tools, etc.) → site activation (initiation visit, training, investigational product readiness) → FPI
Some workstreams can run in parallel. IND preparation can move alongside IRB preparation, and contract/budget discussions can be pulled into the MFDS review window to shorten total duration. Parallelization works best when teams are prepared to finish in parallel—site submission packs, contracts, training plans, and system readiness still need to be in place. When those items are late, the advantage of parallel review disappears.
Related article: Korea clinical trial strategy after MFDS reforms
Practical planning uses ranges—not a single number
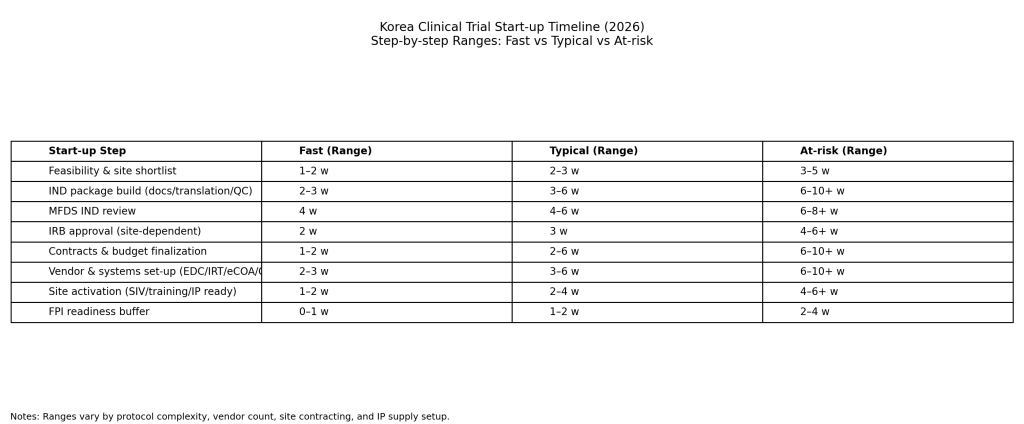
Korea start-up timelines vary by protocol complexity, vendor count, number of sites, central lab/imaging requirements, and investigational product supply model. For real planning, “fast/typical/at-risk” ranges are more stable than a single fixed number.
You can group the start-up clock into five blocks:
Feasibility and site strategy → IND package build and submission → IRB approvals → contracting/budget/vendor/system setup → site activation → FPI
Delays tend to appear in repeatable patterns. Teams often lose time due to document alignment gaps (protocol, ICF, investigator materials, safety flow), incomplete site submission packs, contract/budget back-and-forth, and vendor validation and interface testing. Identifying the main drivers early stabilizes the plan.
Feasibility and site strategy
Feasibility is most useful when it turns assumptions into operational numbers and conditions. This step determines site type, a realistic site shortlist, expected enrollment pace, and operational complexity.
Start with the site model. Will you need major tertiary hospitals, specialist centers, or a network approach? Then account for competitive trials and shifts in standard of care—especially in oncology and rare disease, where competition can change enrollment assumptions quickly. Also stress-test visit burden and window tightness. Heavy schedules and narrow windows often increase missing data and dropouts, which later creates interpretation and reporting work during CSR drafting and global submission packaging.
The most practical outputs from feasibility are: a site shortlist, enrollment assumptions, and a short risk memo highlighting likely bottlenecks.
IND package preparation and MFDS review
The IND timeline typically has two layers: the time to build a complete package, and the MFDS review clock.
Public guidance commonly references an MFDS review benchmark around 30 business days. Operationally, many teams plan for roughly 4–6 weeks, with buffer time if clarification questions or supplementation cycles occur.
In practice, the most common cause of delay is not missing documents—it is misalignment between documents. The protocol, investigator’s brochure, quality-related documentation, safety reporting flow, and dosing/labeling details must connect cleanly. When the “story” changes across documents, review questions increase and schedules extend.
A simple and effective tactic is to maintain a one-page “submission alignment sheet”: key protocol claims and rationale → supporting document name/version → section references → operational implementation notes. This reduces internal churn pre-submission and speeds response handling after submission.
IRB and site submissions
Korea can support parallel regulatory and IRB preparation, but real speed comes from managing the site submission process—not just the IRB meeting calendar.
Many large institutions complete IRB review in roughly a few weeks, but delays often come from site-specific requirements and submission pack completeness. Typical friction points include ICF language (privacy and compensation), recruitment materials, site formatting requirements, and response time for PI/CRC questions.
“First-submission quality” matters. A standardized site submission checklist and fewer rounds of ICF/compensation wording revisions will stabilize the overall start-up schedule.
Contracts, budget, and payment milestones
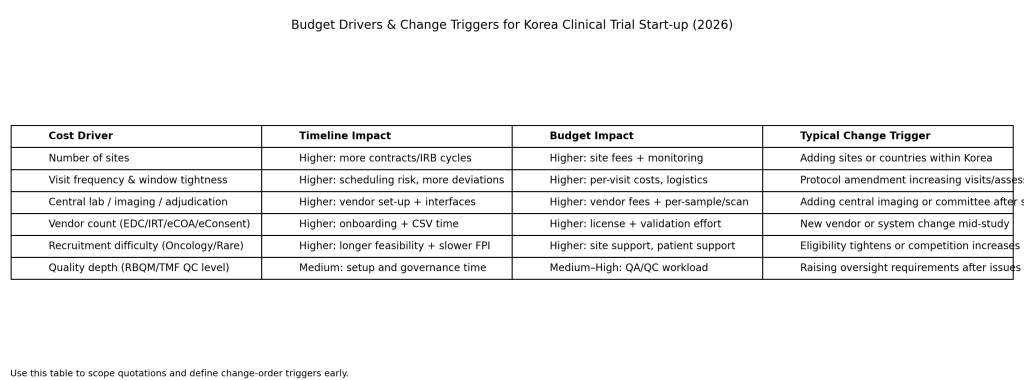
When start-up runs long, contracts and budget alignment are often part of the reason. Schedule stability improves when teams agree early on what drives cost and what triggers a change order.
Typical timeline and budget drivers include: number of sites, visit frequency and window tightness, central lab/central imaging/adjudication needs, number of systems and vendors (electronic outcome tools, randomization and drug supply systems, etc.), recruitment difficulty (oncology/rare disease), and the depth of quality oversight (RBQM intensity and TMF QC depth).
Define change-order triggers up front, and align payment milestones (start-up, activation, enrollment, database lock). This reduces rework and prevents delays caused by cross-border internal approvals and payment logistics.
Vendor and system setup (data capture, randomization/drug supply, electronic outcomes)
Modern trial operations treat vendors as trial system components, not just outsourced services. Early alignment on roles and responsibilities, validation evidence, access control, audit trails, change history, and version update policy reduces schedule shocks later.
Two bottlenecks appear repeatedly: slow onboarding and validation evidence readiness, and slow interface setup (for example, central lab data integration with the main data capture system and safety data reconciliation). Both are harder to fix late. Vendor kickoff and interface test planning should be part of start-up—not something deferred.
Site activation to first patient in
FPI is driven by operational readiness at the site. Key readiness items include: clear training completion criteria, delegation and system access setup, investigational product handling workflows (labeling, storage, accountability, return), and a dry-run of the first visit workflow. If your study involves importation, customs clearance, or labeling constraints, supply chain planning should start earlier—these issues commonly surface late and can delay FPI.
Top 10 causes of start-up delays (and how to prevent them)
In Korea, many delays come from operational work rather than regulatory review itself. Common patterns include: prolonged contract negotiations, repeated translation revisions, slow vendor onboarding/validation readiness, repeated site budget negotiations, multiple rounds of ICF/privacy/compensation edits, delayed central lab/imaging setup, investigational product import/labeling constraints, incomplete training requiring rework, overly optimistic feasibility assumptions leading to slower enrollment, and protocol complexity increasing operational burden.
The best prevention is early structure: align contract templates and payment terms, maintain a glossary and version control discipline, and plan vendor kickoff plus validation pointers early.
Six variables that most affect quotations
Quotation size and timeline frequently move together based on: site count and geographic distribution, visit frequency and window tightness, central lab/imaging/adjudication requirements, number of vendors/systems, recruitment difficulty (oncology/rare disease), and quality oversight depth (RBQM and TMF QC).
When sponsors decide these variables early, CRO quoting becomes faster and change cycles shrink. Even a simple decision—minimum viable setup versus “submission-ready from day one”—meaningfully shifts the plan.
A quick way to calibrate your timeline
A Phase II oncology study with three sites plus central lab and imaging tends to be driven by vendor setup, data interfaces, and site training/standardization. A simpler Phase I or PK-focused study is often driven by site count and visit structure. Identifying which profile matches your protocol helps you place your project into the appropriate timeline band earlier.
Conclusion and next steps
Korea start-up timelines depend on how early the key bottlenecks are addressed—site submission packs, contracts and budget alignment, vendor/system readiness, and supply chain planning. When these items are organized early, schedules stabilize and budget surprises decrease.
Intoinworld is a Korea-based CRO with over 10 years of local execution experience across Phase I–IV and post-marketing studies. We support global sponsors end-to-end, including IND and IRB readiness, site operations, monitoring, data management, and safety operations. If you would like a quotation, please click the Quotation button on this page to request a quote.
FAQ
How many weeks should we plan for a Korea start-up?
Most teams plan within three bands: fast (8–10 weeks), typical (12–16 weeks), and at-risk (16–24+ weeks), depending on protocol complexity and readiness. Contracting, translations, vendor/system setup, and investigational product supply can extend timelines.
Can IND and IRB work run in parallel in Korea?
Parallel preparation is often feasible. The time savings are real when the site submission pack, contracts/budget, and training plans are ready early. If those items lag, the benefit of parallelization decreases.
Where do timelines most commonly slip?
Operational areas tend to drive delays more often than the MFDS review window: contracts and budgets, repeated translation revisions, vendor validation evidence readiness, central lab/imaging setup, and investigational product import/labeling constraints.
What variables affect the budget quote the most?
The largest drivers are site count and distribution, visit frequency and window tightness, central lab/imaging/adjudication requirements, vendor/system count, recruitment difficulty (oncology/rare disease), and the depth of quality oversight (RBQM and TMF QC).
What should we confirm right before FPI?
Confirm training completion and delegation, system access, investigational product workflows (labeling, storage, accountability, return), and a dry-run of the first visit workflow. Supply chain and system readiness are common late-stage delay drivers, so early confirmation is recommended.



