What is Obesity and What Causes It?
Obesity is a chronic condition characterized by excessive fat accumulation, diagnosed when the Body Mass Index (BMI) exceeds 30, with a BMI of 25-29.9 classified as overweight [1]. It contributes to health issues such as type 2 diabetes, cardiovascular diseases, osteoarthritis, and certain cancers [1]. The World Health Organization (WHO) has labeled obesity a global health crisis, with over 900 million adults affected in 2022, representing about one-eighth of the world’s population [2]. Causes include genetic factors, lifestyle habits (e.g., high-calorie diets, lack of exercise), environmental influences (e.g., high-calorie food environments), and psychosocial stress [3]. In some cases, medications or single-gene disorders may be primary triggers, necessitating personalized treatment approaches and evolving therapeutic landscapes.
Recent Trends in Obesity: A Growing Global Challenge
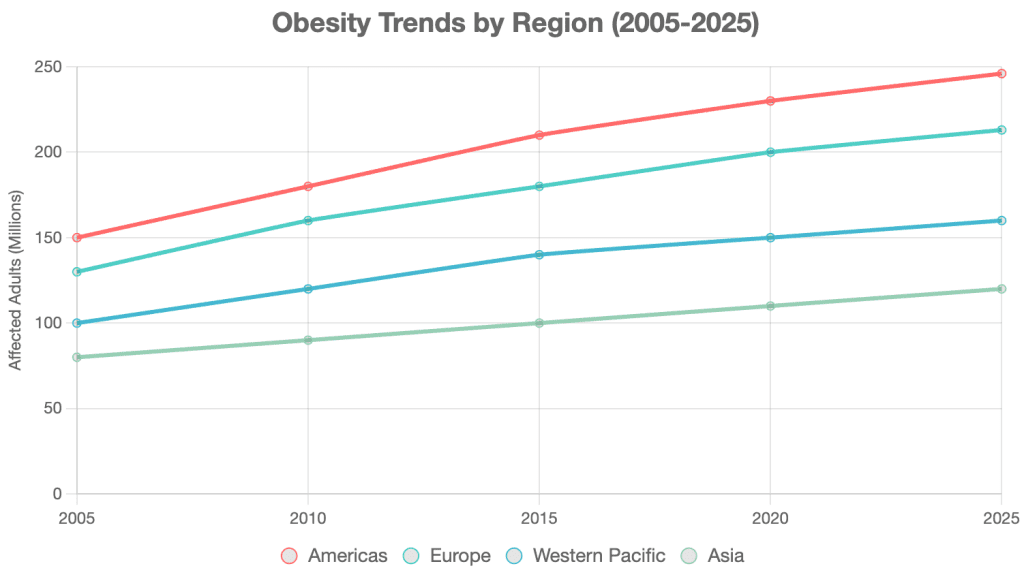
Recent statistics highlight a sharp rise in obesity prevalence. As of 2025:
- Americas: 246 million people
- Europe: 213 million people
- Western Pacific: 160 million people [4]
Southeast Asia and Africa, currently with lower rates, are projected to double by 2035. This indicates obesity is no longer a regional issue but a global public health concern. High-income countries like the U.S., Mexico, the UK, and Australia report the highest prevalence, with middle-income nations also seeing rapid increases [4]. The overweight adult proportion reached 43% in 2022, up from 25% in 1990 [3]. Consequently, the obesity treatment market is booming, with a projected Compound Annual Growth Rate (CAGR) of 22.31% from 2025 to 2030, reaching $605.3 billion by 2030 [5].
Advances in Obesity Drug Development
Obesity drug development has seen remarkable progress through innovative therapies. Notable GLP-1 receptor agonists, semaglutide and tirzepatide, achieve 15-20% average weight loss and improve cardiovascular and metabolic health [6]. Guidelines recommend these drugs for BMI ≥30 or BMI ≥27 with comorbidities [4]. Emerging treatments include:
- Dual/triple agonists (GLP-1/GIP): Enhanced weight loss efficacy
- MGAT2 inhibitors, ACTR2 antagonists: Targeting GLP-1-resistant patients
- Gene therapy, microbiome interventions: Diversifying treatment options
- Biomarker-based therapies (leptin, adiponectin, CRP, IL-6) [7]
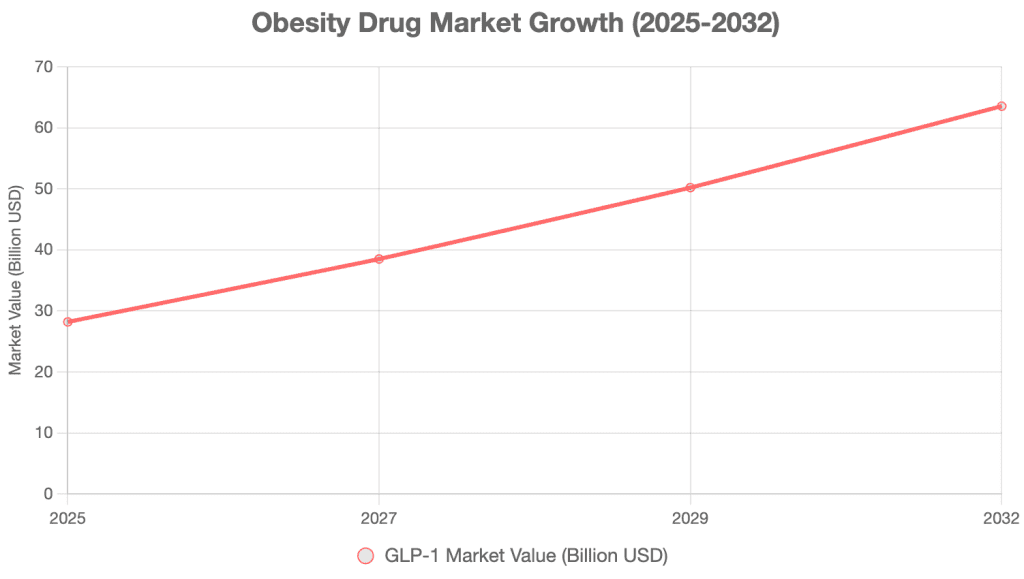
The global GLP-1 market is expected to grow from $281.9 billion in 2025 to $635.4 billion by 2032, with a CAGR of 12.3% [8].
Evolution of Obesity Clinical Trials
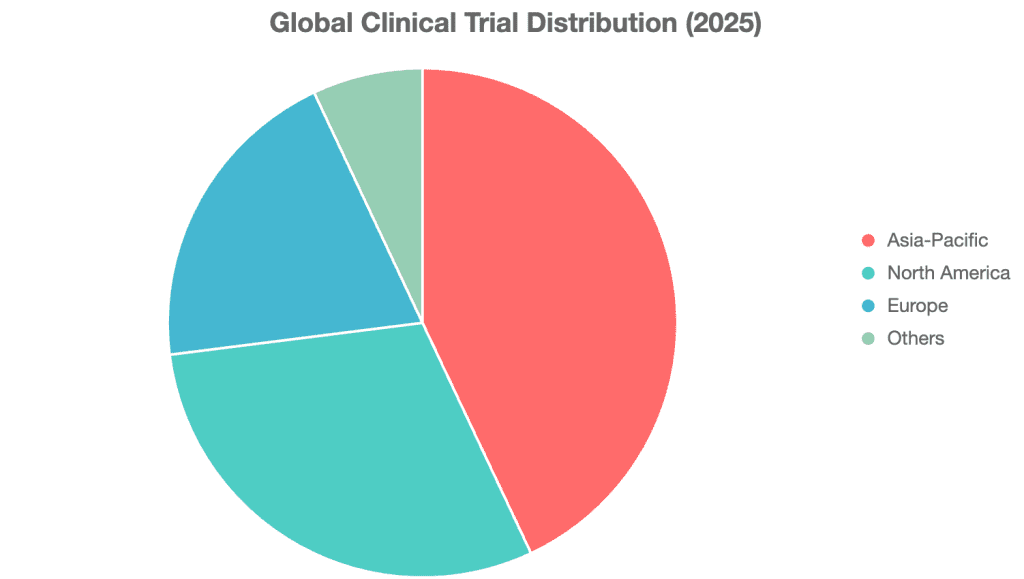
Obesity-related clinical trials are expanding rapidly. Since 2019, over 1,400 trials have been initiated, with an annual growth rate of approximately 20% [9]. Regionally:
- Asia-Pacific: 43% of total trials, leading the field
- North America: ~30%
- Europe: ~20% [9]
Mainland China and the U.S. are the most active, while Germany, Korea, and Australia excel in early-phase (Phase I/II) trials. The pipeline includes over 120 weight-loss drugs from 60 companies, signaling intense competition [10].
Why South Korea Ranks as the 4th Top Medical System Country for Obesity Clinical Trials
Korea ranks 4th globally in clinical trials in 2025, underpinned by its world-class healthcare system and 97% national health insurance coverage [11]. Key strengths include:
- MFDS-designated trial sites: Over 220
- Dedicated Phase I units: 28
- Research beds: Over 750
- Seoul: #1 in trial volume since 2017 [11]
Additionally:
- FDA inspection report “Official Action Indicated (OAI)”: 0 cases from 2008-2022
- MFDS IND approval time: Average 30 days (notification within 90 days)
- IRB review time: Average 3 weeks
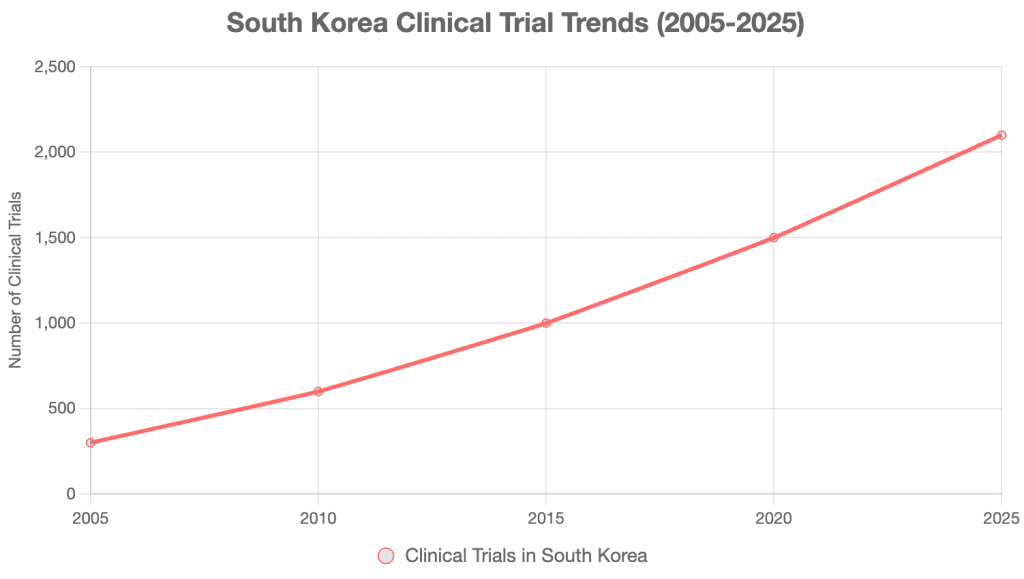
Korea’s clinical trial numbers have grown from 300 in 2005 to 2,100 in 2025, a 600% increase [11].
Implement Obesity Solutions with Intoinworld
If you are looking for ways to reduce clinical trial costs, we encourage you to request a quote from Intoinworld to receive a tailored consultation. And if you don’t want to miss the latest pharma/biotech industry trends and clinical trial insights, consider subscribing to Intoinworld’s newsletter to get valuable information on a regular basis.
References
[1] WHO, “Obesity and Overweight”, https://www.who.int/health-topics/obesity
[2] WHO, “Obesity and Health Risks” (2023), https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight
[3] WHO, “Causes of Obesity” (2023), https://www.who.int/news-room/q-a-detail/obesity-causes
[4] Global Health Observatory, “Obesity Prevalence Data (2025 Projections)”, https://www.who.int/data/gho/data/themes/topics/topic-details/GHO/obesity
[5] Market Research Report, “Obesity Treatment Market Forecast, 2030”, https://www.grandviewresearch.com/industry-analysis/obesity-treatment-market
[6] International Diabetes Federation, “GLP-1 Agonists in Obesity Management”, https://www.idf.org/glp-1-obesity
[7] Emerging Therapies Journal, “Innovative Obesity Treatments” (2025), https://www.emergingtherapiesjournal.org/obesity-treatments
[8] Pharmaceutical Market Insights, “GLP-1 Market Projections, 2032”, https://www.pharmainsights.com/glp-1-market
[9] Clinical Trials Database, “Global Obesity Trial Trends”, https://clinicaltrials.gov/obesity-trends
[10] Industry Analysis Report, “Obesity Drug Pipeline”, https://www.iqvia.com/obesity-pipeline
[11] Ministry of Food and Drug Safety (MFDS), “South Korea Clinical Trial Statistics (2005-2025)”, https://www.mfds.go.kr/clinical-trial-stats
[12] MFDS, “Clinical Trial Authorization Guidelines”, https://www.mfds.go.kr/cta-guidelines

