South Korea’s global reputation, fueled by cultural phenomena like K-POP, cutting-edge technology, and K-brands, is now extending into the realm of pharmaceuticals. The country has rapidly become a Clinical Trials powerhouse, drawing attention from international sponsors. In just a few years, Korea’s clinical research landscape has transformed – backed by striking growth statistics and a strong foundation in infrastructure and regulation. According to the MFDS (Ministry of Food and Drug Safety), the number of approved Clinical Trials in Korea jumped from 679 in 2018 to approximately 1,723 in 2023 – over a 150% increase in five years. This dramatic rise is driven by advanced medical infrastructure, a diverse patient pool, and an efficient regulatory environment that together make Korea an attractive hub for Clinical Trials.

Korea’s Clinical Trial Environment: Key Metrics
To understand Korea’s appeal in Clinical Trials, consider its key metrics and global rankings. The nation of ~51.6 million people boasts one of the world’s top-rated healthcare systems and virtually universal health coverage (97% of the population). Its pharmaceutical market is the 11th–13th largest globally, underpinned by strong R&D investment. Crucially, the MFDS – Korea’s regulatory agency equivalent to the FDA – is known for speedy approvals, often reviewing IND applications within 30 working days on average. Institutional Review Board (IRB) ethics approvals at hospitals take only about 3 weeks on average. Seoul, the capital, has been the #1 city globally for number of Clinical Trials since 2017. Over 200 hospitals and medical centers have been MFDS-designated as qualified clinical trial sites, with more than half located in Seoul and surrounding regions. Korea also has 26–28 dedicated Phase I clinical trial units for early-phase research – among the highest density in the world. Notably, from 2008 to 2022, U.S. FDA inspections in Korea resulted in zero Official Action Indicated (OAI) warnings, attesting to the high quality and compliance of Korean trial sites.

| Key Indicator | Korea’s Clinical Trials Profile |
| Population (2025) | ~51.6 million (with 97% under national health insurance) |
| Global Pharma Market Rank | Top 12 globally (by market size) |
| Healthcare System Ranking | Among world’s best (OECD assessments, 2025) |
| MFDS IND Approval Timeline | ~30 working days (IND approval median), fast-track options available |
| IRB Ethics Approval Timeline | ~3 weeks on average (site-level IRB review) |
| Global Clinical Trial Rank | Top 5 country for clinical trial activity |
| MFDS-Designated Trial Sites | 220+ certified hospitals (≈ half in Seoul metro) |
| Seoul’s Ranking by Trial Count | #1 city globally for trials since 2017 |
| Dedicated Phase I Units | 26 (independent early-phase clinical pharmacology centers) |
| FDA Inspection Track Record | 0 OAI warnings (2008–2022) (exemplary compliance) |
| Clinical Research Beds | 700+ dedicated beds for studies (in top hospitals) |
These indicators highlight why Korea’s Clinical Trials environment stands out. A large, insured population means faster patient recruitment for trials, and dense urban hospital networks enable efficient multi-center studies. The MFDS’s streamlined processes and global-standard regulations ensure that trials in Korea meet FDA/EMA requirements, allowing Korean trial data to be used for international submissions. In fact, Korea’s membership in the International Council for Harmonization (ICH) and status as a WHO Listed Authority (WLA) mean that well-conducted Clinical Trials in Korea are recognized by regulators in the US, EU, and beyond. This global acceptance of Korean data is a major draw for sponsors looking to run trials that can support approvals in multiple regions with one study.
Global Competitiveness and Rapid Growth
Korea’s rise in clinical research is reflected in global rankings. In 2023, South Korea ranked fourth worldwide for clinical trial activity, even as overall global trial numbers dipped by 5.5% that year. Seoul remains the busiest city for Clinical Trials on the planet. According to the Korean National Enterprise for Clinical Trials (KoNECT), while other countries saw slowdown, Korea’s trial activity increased by 9% in 2023. This momentum has been building over the past decade: South Korea has been among the top 10 countries for Clinical Trials since 2010, and is especially renowned for oncology and early-phase research. By one analysis, Korean investigators now conduct about 3.5% of all global industry-sponsored Clinical Trials, up from virtually nothing two decades ago.

Importantly, Korea is not just doing a large volume of trials – it’s doing high-quality trials. The country places strong emphasis on Good Clinical Practice (ICH-GCP) compliance and data integrity. All major university hospitals have dedicated Clinical Trial Centers with internal audit teams to ensure studies meet international standards. ClinicalTrials.gov data also show Korea excels in Investigator-Initiated Trials (IITs), ranking among the top 5 globally for researcher-led studies. This indicates a robust academic research culture and the availability of experienced investigators. Taken together, these factors position Korea as a strategic clinical trial hub where global pharma and biotech companies are increasingly investing.

World-Class Hospitals and Infrastructure
Another reason Korea is a hotspot for Clinical Trials is its concentration of world-class hospitals and research infrastructure. The MFDS has certified over 220 hospitals as qualified clinical trial sites, ensuring they meet stringent standards for conducting trials. Notably, more than half of these are located in Seoul and the surrounding Gyeonggi Province, enabling efficient multi-center Clinical Trials with minimal logistical hurdles (many sites can be reached within an hour’s drive of each other). This dense network makes Korea ideal for studies requiring large patient enrollment from multiple hospitals.
Top 10 Clinical Trial Hospitals in Korea: (MFDS-designated leading sites)
- Samsung Medical Center (Seoul) – Tertiary hospital with cutting-edge research facilities
- Asan Medical Center (Seoul) – One of Asia’s largest hospitals, renowned for oncology trials
- Seoul National University Hospital (Seoul) – Prestigious academic medical center, top-tier in trials
- Seoul St. Mary’s Hospital – Catholic Univ. (Seoul) – Major trial center with international accreditations
- Yonsei Severance Hospital (Seoul) – Historic institution known for advanced trials in multiple fields
- National Cancer Center (Ilsan, Gyeonggi) – Specialized in oncology Clinical Trials and research
- SNU Bundang Hospital (Seongnam) – High-tech hospital in metro Seoul with active trial units
- Chonnam National University Hwasun Hospital (Jeollanam-do) – Leading regional cancer trial center
- Korea Institute of Radiological & Medical Sciences (Seoul) – Specialized in radiology and oncology trials
- Korea University Guro Hospital (Seoul) – Large academic hospital engaged in diverse Clinical Trials
These institutions feature state-of-the-art equipment, experienced research personnel, and adherence to international GCP standards. Many have dedicated Clinical Trial Units or Centers that coordinate studies, some with Phase I clinical pharmacology wards and inpatient beds specifically for trial participants. The high quality of data from these hospitals is evidenced by the lack of critical findings from foreign inspections; for example, no FDA official action indications in 15+ years. Additionally, Korea’s widespread adoption of Electronic Medical Records (EMR) across hospitals facilitates efficient patient data management and recruitment for Clinical Trials. Investigators can rapidly identify eligible patients through EMR databases while ensuring data accuracy and compliance.
Moreover, K-Health infrastructure – much like K-POP in culture – has become a strong “brand.” International sponsors recognize names like Samsung Medical Center or SNU Hospital for their excellence. This brand trust, combined with English-speaking coordinators and globally experienced investigators, gives sponsors confidence in choosing Korea for their trials. As a result, Korea routinely collaborates on multinational trials funded by global pharma. In 2018, for instance, 289 multinational studies were conducted in Korea (out of 679 total trials that year), and this proportion has only grown since. Korean sites are often among the top enrollers in global studies, especially in oncology, reflecting both the patient availability and the clinician expertise.
Thriving Biotech Market and “K-Bio” Momentum
Beyond infrastructure, South Korea’s booming biotech and pharmaceutical market makes it an attractive place for Clinical Trials. Often dubbed “K-Bio” by media, the sector’s rise mirrors the global craze for K-culture. After K-pop, K-dramas, and K-beauty, Korea is now turning its focus to biotechnology. The domestic biopharma market is valued around $22–25 billion (13th largest globally) and is projected to continue robust growth. Government initiatives and incentives play a big role: the Korean government set a vision for “K-Bio Pharma Global Top 5” and launched a $19 billion R&D plan to fuel biotech startups and innovation. By 2030, Korea aims to rank among the top six pharmaceutical powerhouses.
This pro-innovation climate has led to a surge in new drug development and, consequently, Clinical Trials to test those innovations. In fact, Korea now ranks third worldwide for new drug discovery output, with over 1,300 new drug candidates generated by Korean companies in the last three years. Many of these novel therapies – from cell and gene therapies to novel oncology drugs – enter Clinical Trials in Korea first. The pipeline vitality ensures a steady stream of trials sponsored by domestic firms, adding to those sponsored by multinationals.
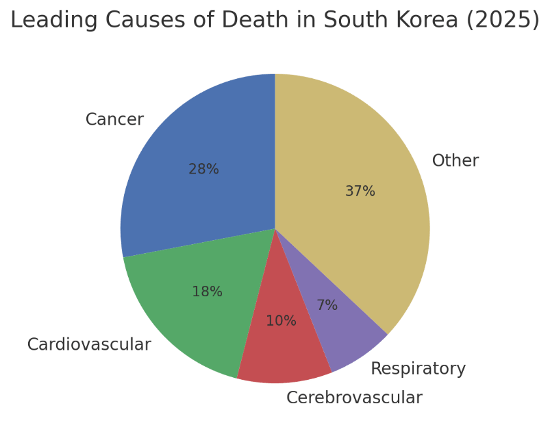
Investors have noticed. Global pharma companies increased R&D investment in Korean clinical research by ~6.7% in 2023, spending roughly ₩873 billion (~$640 million) that year. Over 69% of major trials in Korea in 2023 were in cancer – a priority area given Korea’s epidemiology – and around 13.5% targeted rare diseases. Korea’s high incidence of certain cancers (cancer is the leading cause of death at 28%) makes it a strategic location for oncology trials, as patients and specialist investigators are readily available. Cardiovascular disease is another high-impact area (18% of deaths), reflected in many cardio-metabolic trials. These factors align the Clinical Trials market with public health needs, often leading to faster enrollment and meaningful outcomes.
Economically, the growth of the clinical trial sector has significant impacts. Korea’s domestic pharmaceutical sales have been climbing sharply; by one estimate, they could reach ₩44.6 trillion by 2025, roughly doubling from 2019 levels. This is buoyed by factors like increased healthcare spending, a rise in drug exports (up 15% recently), and regulatory reforms that encourage innovation. The MFDS’s introduction of programs like GIFT (Global Innovative Fund for Technology) and fast-track designations, as well as alignment with FDA’s Breakthrough Therapy Designation, have streamlined development of cutting-edge drugs. Additionally, the government’s “Bioeconomy” initiatives and tax incentives have attracted significant foreign direct investment into Korean biotech. Major global deals – such as GSK’s £2 billion partnership with Korea’s ABL Bio in 2025 – underscore the international confidence in Korean biotech. All these trends create a virtuous cycle: a thriving Korean biotech industry draws more trials, and successful Clinical Trials further strengthen the industry’s global credibility.
Regulatory Excellence and MFDS Efficiency
Korea’s efficient and innovative regulatory framework is a cornerstone of its appeal for Clinical Trials. The MFDS has continually updated its processes to be faster and more sponsor-friendly. In early 2025, the MFDS implemented sweeping drug approval reforms, introducing dedicated review teams, rolling submissions, and parallel reviews to cut new drug approval times from ~420 days to 295 days. These changes signal to sponsors that Korea is serious about being a strategic clinical trial hub, not just another participating country. For clinical trial applications specifically, the MFDS allows sponsors to file the trial protocol for approval in parallel with IRB ethics submissions, saving time. Sponsors typically receive MFDS trial approval in about 4–8 weeks, faster than in many other countries. (For comparison, regulatory approval can take 10–12 weeks in Japan and 12–16 weeks in China.) This speed does not come at the expense of rigor – it’s backed by highly qualified reviewers and a transparent process with opportunities for sponsor input (the MFDS grants up to 10 consultation meetings during a new drug review, for example).
Another advantage is Korea’s embrace of electronic submissions and harmonized standards. As an ICH member, MFDS accepts Common Technical Document (CTD) formats and adheres to GCP and data integrity guidelines like the FDA and EMA do. In 2022, the World Health Organization recognized MFDS as a “Level 4” maturity agency (WLA), meaning its oversight is on par with top regulators like FDA. This gives foreign sponsors confidence that trials conducted in Korea won’t face regulatory snags. Indeed, under FDA’s 21 CFR 312.120 and equivalent EMA guidelines, data from foreign sites (like Korea) can be used in applications as long as GCP is followed– a criterion Korea meets consistently. Many sponsors have leveraged this by running a single pivotal Clinical Trial in Korea to support drug approvals in multiple markets. For instance, a well-designed Korean trial can potentially be submitted to MFDS, FDA, and EMA simultaneously without duplicate studies, accelerating global development timelines.
MFDS also offers regulatory innovations such as the GIFT program and Priority Review for certain critical therapies. These programs can expedite Clinical Trials for orphan diseases, serious conditions, or innovative treatments by providing extra support and faster review. Moreover, Korea’s regulatory environment is highly transparent – guidelines and review timelines are clearly published, and the MFDS frequently updates sponsors on new regulations (such as digital health device trial guidelines in 2025, etc.). The end result is a regulatory climate that balances speed and quality, which international sponsors find very appealing when planning trials.

Expansion of CROs and Operational Support
A critical element of Korea’s clinical trial boom is the expansion of its Contract Research Organization (CRO) industry. As more trials come to Korea, a robust ecosystem of CROs – both global and local – has grown to support sponsors with on-the-ground execution. Approximately 65 CRO companies now operate in Korea, providing services ranging from regulatory consulting and site management to data analysis. Over the past few years, this sector has shown dramatic growth: the Korea CRO industry’s workforce grew by 47.4% in the last 3 years, and CRO revenue in Korea jumped 77.7% over the past 5 years. This indicates both rising demand and increasing capacity for handling Clinical Trials.
Global CRO giants (IQVIA, Parexel, ICON, etc.) hold roughly 70% of the Korean market share, leveraging their international experience to win projects. Meanwhile, 20–30 local CROs compete with strengths in local expertise, relationships, and cost-effectiveness. Sponsors have ample choice in finding a CRO in Korea that fits their trial’s needs. Many domestic CROs specialize in certain therapeutic areas (e.g., oncology or medical devices) and offer tailored services with lower overhead costs. Clinical Trials in Korea often benefit from lower operational costs overall – estimates suggest trial costs in Korea are 30–40% lower than in the United States for equivalent quality. For example, a global Phase 3 diabetes trial that partnered with a Korean CRO was able to reduce costs by 35% and shorten the study timeline by 6 months, compared to initial projections.
The presence of experienced CROs also helps new sponsors navigate Korea’s environment smoothly. CROs facilitate rapid site startup by knowing the local IRB processes, guiding sponsors through MFDS documentation, and coordinating patient recruitment using local networks. They also ensure language is not a barrier – most Korean investigators and study staff are proficient in English, but having bilingual CRO coordinators streamlines communications. Furthermore, Korean CROs maintain high quality standards; they operate under ICH-GCP and have internal QA systems, which is evident from the consistently clean FDA inspection record in Korea.
Overall, the CRO expansion means that even sponsors without a physical presence in Korea can run trials here effectively by outsourcing to capable CRO partners. The result is a well-oiled machine: sponsors bring protocols, CROs and hospitals in Korea deliver results. With such support, it’s no surprise that South Korea is rapidly emerging as a global hub for Clinical Trials, combining K-brand prestige with scientific prowess. (If you are planning a study, Intoinworld offers a Korea CRO selection guide outlining key strategies to ensure trial success with the right partner.)
Work with Intoinworld

If you are considering conducting Clinical Trials in Korea and want expert guidance, use our Quotation Request page to get a tailored proposal. Intoinworld’s team – with over 10 years of CRO experience across Phases I–IV and post-market studies – will consult with you to design a strategic plan aligned with MFDS regulations and your project goals.

Stay informed on the latest Clinical Trials trends, MFDS regulatory updates, and success stories from the Korean biotech industry by subscribing to Intoinworld’s newsletter. Our regular updates will help you navigate Korea’s clinical trial environment with insider knowledge. Sign up to receive expert insights delivered to your inbox.
FAQ
Q1. Why is South Korea becoming a hotspot for Clinical Trials?
A1. South Korea offers a unique combination of fast regulatory approval, world-class hospitals, and strong patient enrollment capabilities. The MFDS provides efficient trial approval (often within 30 days) and adheres to international GCP standards, meaning Korean trial data is accepted by the FDA and EMA. Korea also has over 220 certified trial sites and a 97% insured population, enabling quick recruitment for Clinical Trials. Additionally, the country’s global reputation (bolstered by “K-” cultural influence) has increased foreign sponsor interest. All these factors make Korea an attractive and credible hub for Clinical Trials.
Q2. What advantages do Korean hospitals offer for Clinical Trials?
A2. Korean hospitals provide advanced infrastructure and experienced research teams. Many large hospitals in Seoul – such as Samsung Medical Center and Seoul National University Hospital – have dedicated clinical research centers and vast patient databases. They can conduct complex trials (e.g. oncology, immunotherapy, medical devices) with high quality. Electronic Health Records and centralized healthcare in Korea help identify eligible patients faster. Moreover, Korean sites have an excellent compliance record (no major findings in FDA inspections). Sponsors benefit from reliable data and often faster enrollment at Korean hospitals, which consistently rank among the top globally for trial activity.
Q3. How does the MFDS ensure fast yet rigorous approval of Clinical Trials?
A3. The MFDS (Ministry of Food and Drug Safety) has implemented streamlined processes for clinical trial approvals. Sponsors can submit trial applications in parallel to the MFDS and institutional IRBs, saving time. The MFDS typically gives an IND approval or feedback within about 4–8 weeks. They have dedicated review teams that can engage in sponsor consultations to clarify requirements. While fast, the process is rigorous – MFDS requires trials to meet ICH-GCP standards and often asks for additional information if something is unclear. Korea’s membership in ICH and recognition as a WHO Level 4 regulatory authority reflects its high standards. Thus, Clinical Trials get approved quickly without compromising on quality, and the outcomes are trusted globally.
Q4. What is the role of CROs in Korea’s Clinical Trials industry?
A4. Contract Research Organizations (CROs) in Korea are key facilitators that help plan and execute trials for sponsors. They handle local regulatory submissions, site coordination, patient recruitment, and data management. Korea’s CRO industry has expanded rapidly – with around 65 CROs now, including global firms and local specialists. CROs provide local expertise, language support, and operational efficiency, making it easier for foreign sponsors to run trials without a direct presence. They also contribute to cost-effectiveness: conducting Clinical Trials in Korea through a CRO can be 30–40% cheaper than in Western countries for comparable quality. By choosing the right CRO partner, sponsors benefit from the CRO’s experience with Korean sites, understanding of MFDS procedures, and network of investigators, ensuring smoother trial conduct.
Q5. Are results from Korean Clinical Trials accepted by the FDA and EMA?
A5. Yes. Data from well-conducted Korean Clinical Trials are accepted by major regulators like the U.S. FDA and European EMA, as long as the trials adhere to GCP and regulatory guidelines. South Korea follows ICH guidelines, and the MFDS is a recognized stringent regulatory authority (a WHO-designated “benchmark” agency). There have been many cases where Korean trial data supported FDA approvals or were included in global new drug applications. Sponsors often design Korean trials to meet global standards; for instance, endpoints and protocols can be aligned with FDA requirements. As a result, a single Clinical Trial in Korea can potentially support drug approval submissions in multiple markets, which is a huge advantage in accelerating development timelines and avoiding redundant trials.

