Introduction – ADC clinical trials go mainstream
Antibody‑drug conjugates have moved from the fringes of oncology to the centre of many treatment algorithms. By 2026, HER2‑low and TROP‑2 biomarkers significantly expand the pool of patients eligible for ADC clinical trials while adding complexity to every aspect of planning. Sponsors and clinical research management teams must decide how to define and measure these biomarkers, choose between basket designs or tumour‑specific programmers, and select regions that can support multi‑year oncology medical trials. In this context, korea clinical trial capabilities and collaboration with a clinical research organization become strategic decisions.

Why Korea is Becoming a Strategic Hub
How HER2‑Low Changes Trial Design
HER2‑low redefines HER2 as a spectrum rather than a binary. Including this group enlarges the potential participant population, but only if pathology assessments are consistent. Sponsors must balance rapid local testing with central review to avoid variability. They also need to decide whether to test their ADCs in late‑line settings first or move earlier into the treatment algorithm, which requires longer follow‑up and more rigorous comparators.
TROP‑2 and Multi‑Tumour Platforms
TROP‑2 expression across breast, lung, gynaecologic and gastrointestinal cancers encourages platform strategies. Sponsors can pursue indication‑by‑indication development or design basket trials that cross tumour types. Each option has advantages and challenges: sequential programmes align with standard care but take longer, while basket trials are efficient but complex to analyse. In a competitive environment, operational excellence—patient selection, safety management, clean data—is a key differentiator.
Chart: Growth of HER2‑Low and TROP‑2 ADC Trials
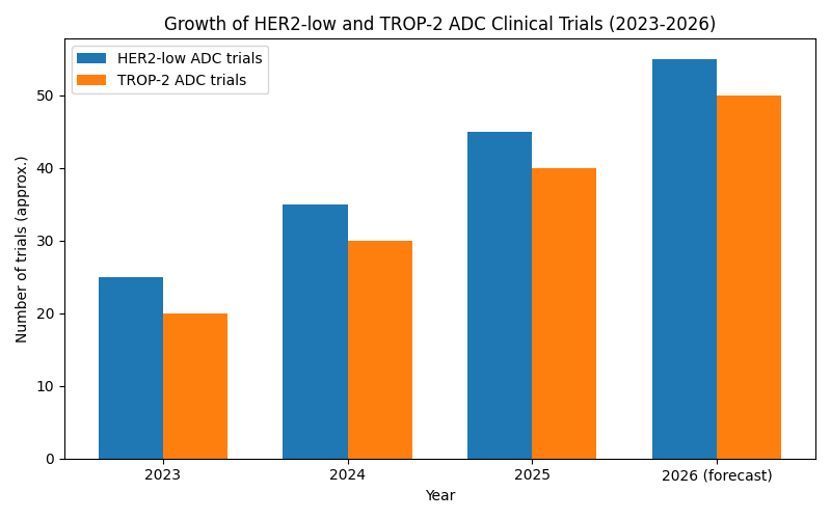
The chart illustrates hypothetical growth in the number of ADC trials targeting HER2‑low and TROP‑2. Both lines are forecast to rise through 2026 as these programmes become mainstream.
Chart: Shift from Late‑line to Early‑line Use
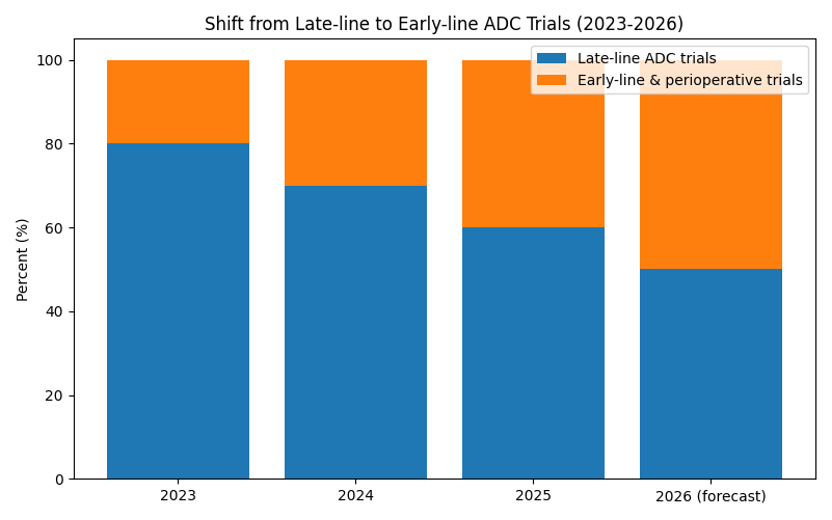
ADC trials are moving from late‑line settings into earlier stages and perioperative contexts. The stacked bar chart shows how late‑line trials shrink while early‑line and perioperative trials expand through 2026.
Table: Comparing HER2‑Low vs TROP‑2 ADC Trials
| Factor | HER2‑Low ADC trials | TROP‑2 ADC trials |
| Target tumours | Mainly breast and gastric cancers with varying HER2 expression | Breast, lung, gynaecologic and gastrointestinal tumours |
| Eligibility criteria | HER2 IHC 1+ or 2+; consistent pathology scoring | TROP‑2 expression (high or intermediate), co‑mutations |
| Operational challenges | Balancing local vs central testing, re‑biopsy logistics | Crowded competitive landscape, statistical complexity of basket designs |
| Development strategy | Stepwise move from late‑line to early lines; combination therapies | Indication‑by‑indication plus basket trials running in parallel |
Chart: ADC Trial Share by Region (2025)
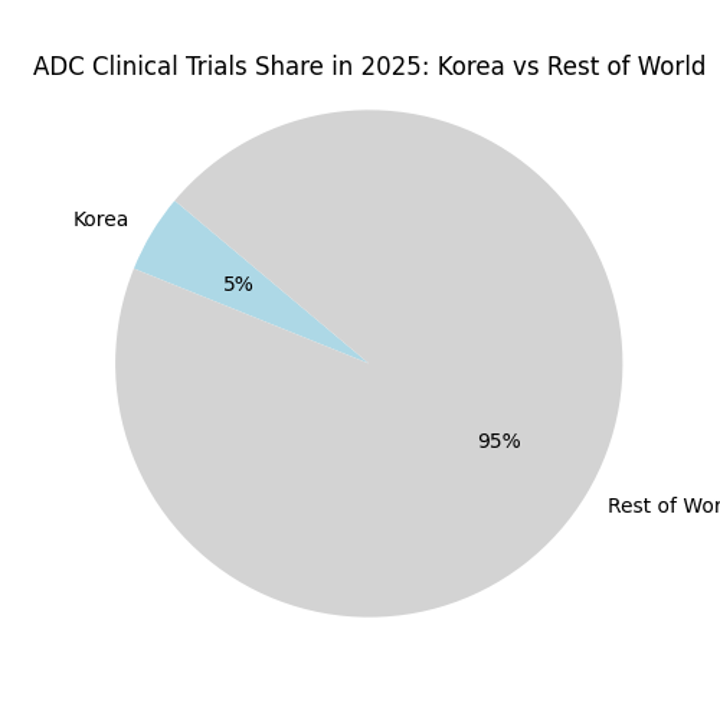
While Korea currently hosts a small share of global ADC trials, reforms such as dedicated MFDS review teams and rolling submission reduce approval timelines and increase data reliability. These factors, combined with the country’s high‑volume cancer centres and unified health system, make Korea an attractive site for ADC programmes.
Why ADC Trials in Korea Matter
ADC programmes require biomarker‑driven recruitment, long‑term safety monitoring and stable regulatory processes. Korea offers high‑throughput oncology centres experienced in HER2 and TROP‑2 testing, a national health insurance system that enables long‑term follow‑up, and regulatory reforms that shorten approval timelines. Clinical research management teams can thus align Korean trial activities with FDA and EMA submissions. Choosing Korea is not only about patient numbers; it is about data quality and operational predictability.
Intoinworld’s Contribution
Intoinworld, a Korea‑based clinical research organization, helps overseas sponsors translate ADC strategies into executable Korean studies. For HER2‑low and TROP‑2 programmes, it works with investigators to align inclusion criteria with local pathology practice, adjust visit and testing schedules to real clinic operations and identify centres with strong oncology expertise. By implementing five pillars—data governance, eCTD mapping, label concordance, inspection & quality bridge and transfer‑package composition—Intoinworld ensures that data generated in Korea can be seamlessly integrated into global dossiers.
Request a Quote and Subscribe to the Newsletter
Request a Project Assessment: Planning to include Korea in your HER2‑low or TROP‑2 ADC programme? Contact Intoinworld via the quotation request form.
Subscribe to the Newsletter: Stay updated on MFDS reforms and clinical research trends by subscribing to Intoinworld’s newsletter. Regular insights and case studies will arrive directly in your inbox.