Introduction: Why South Korea for Radiopharmaceutical Clinical Trials
South Korea has rapidly become a preferred hub for radiopharmaceutical and theranostic Clinical Trials. Thanks to the country’s fast and globally-aligned regulatory system and cutting-edge medical infrastructure, sponsors can initiate studies in as little as 6–8 weeks. The Ministry of Food and Drug Safety (MFDS) provides one of the world’s speediest approval processes – IND approvals often take just 30 working days (~4–6 weeks). Crucially, hospital Institutional Review Boards (IRBs) review ethics in parallel, so total startup time for a Clinical Trial can be under two months.
Beyond speed, South Korea offers internationally recognized quality: as an ICH member, Korea enforces GCP standards equivalent to EU/US, and MFDS-approved trial data is accepted by FDA and EMA. This strong foundation makes Korea ideal for Clinical Trials of novel theranostic radiopharmaceuticals. In this article, we explore how to design and operate successful radiotheranostic Clinical Trials in Korea – focusing on MFDS IND submission, isotope supply logistics, CMC strategy, radiation safety, and Korea’s role in global theranostic development.
MFDS IND Submission for Radiopharmaceutical Clinical Trials
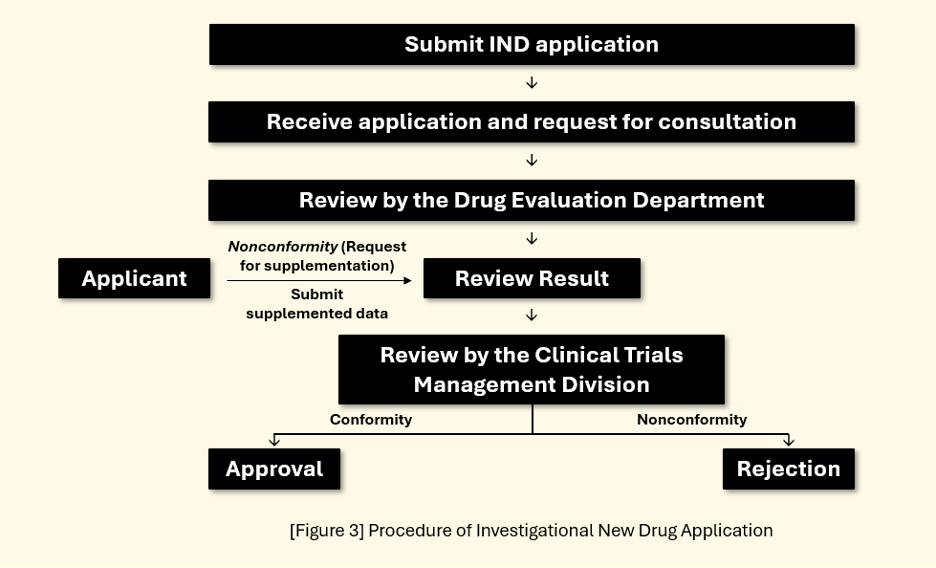
Designing a radiopharmaceutical Clinical Trial in Korea starts with navigating the MFDS IND (Investigational New Drug) application process. MFDS IND requirements closely mirror FDA/EMA standards, but with some local specifics. Sponsors (or their local Korean representative) must submit a trial plan to MFDS and receive IND approval before starting any Clinical Trials. The IND dossier follows ICH Common Technical Document format – including protocol, Investigator’s Brochure, CMC data, and nonclinical results – and must demonstrate compliance with GMP standards.
Notably, for radiopharmaceuticals the MFDS explicitly requires proof that the investigational product is manufactured under GMP for Radiopharmaceuticals (Annex 3-2). This means your CMC section should include documentation of adherence to MFDS’s radiopharmaceutical manufacturing guidelines. Ensuring your isotope labeling, sterility, stability, and purity data meet these standards is critical.
Regulatory timeline: MFDS’s IND review is officially 30 days and often faster in practice. Sponsors can also request a pre-IND meeting to discuss any Korea-specific concerns (e.g. need for local data or dose calibration), which can smooth the review. In parallel, IRB approvals at chosen Korean hospitals should be prepared – these can be submitted during MFDS review to compress timelines. By leveraging Korea’s parallel review system, many radiotheranostic Clinical Trials achieve “First-Patient-In” within ~2 months of IND submission. This agility far outpaces most countries and is a major draw for global sponsors.
Local representation: Foreign sponsors must appoint a local entity or CRO to act as the trial sponsor in Korea. An experienced Korean CRO can handle submissions and communications with MFDS, ensuring nothing is lost in translation. They will prepare Korean-language trial documents (required for protocol, consent forms, etc.) and coordinate with sites. Engaging a local CRO with radiopharmaceutical trial experience is highly recommended to navigate MFDS regulations and operational nuances.
Isotope Logistics and Supply Chain
Conducting theranostic Clinical Trials involves unique isotope logistics challenges. Short half-lives of radionuclides (from hours to days) mean sponsors must carefully plan production, import, and on-site handling of radiotracers. South Korea’s advantage is an integrated hospital-radiopharmacy system and improving domestic isotope supply. Many large Korean trial sites have on-site radiopharmacies or generator facilities, enabling “just-in-time” compounding of tracers under strict quality controls. For example, ^68Ga or ^177Lu labeling can often be done at the hospital just before patient dosing, minimizing decay loss.
If importing radioisotopes or cold kits, sponsors should account for customs clearance and transport time – fortunately, Korea’s import process for trial materials is efficient, but a Radioisotope Import Permit may be needed for certain isotopes. Coordination with the Korea Institute of Radiological & Medical Sciences (KIRAMS) or Korea Atomic Energy Research Institute (KAERI) can help secure supply. In fact, Korea is actively boosting local isotope production: by 2025, domestic production of key isotopes like Actinium-225 and Iodine-131 will reduce reliance on imports.
Government initiatives have established cyclotron facilities to produce Ac-225 for alpha-emitter therapies, accelerating theranostic development. A new consortium of KIRAMS and Korean pharma companies is driving alpha-emitter R&D and proposing regulatory improvements. These efforts mean radiopharma sponsors in Korea can expect a more stable isotope supply chain ahead, including local sources for clinical trial doses.
Global coordination: Sponsors running multi-country radiopharmaceutical Clinical Trials often include Korea alongside other hubs (e.g. Australia) to mitigate supply risks. Because production and delivery are time-critical, spreading sites across regions ensures redundancy. Australia offers fast startup and flexible radiopharmacy capabilities, while Korea contributes advanced imaging and rapid enrollment – together easing radiopharma trial bottlenecks. When designing your trial, consider a supply chain map aligning isotope production schedules with patient dosing across all regions. Build in contingency plans for any shipment delays (e.g. backup manufacturing slots or dose sharing between sites). By leveraging Korea’s growing isotope infrastructure and complementary partnerships, sponsors can greatly stabilize their theranostic Clinical Trials timelines.
CMC Strategy for Theranostic Products (Chemistry, Manufacturing, Controls)
Radiotheranostic products combine a targeting molecule with a radioactive isotope – introducing complex CMC considerations for Clinical Trials. In Korea, MFDS reviewers will scrutinize CMC sections to ensure patient safety and product consistency. Key focus areas include:
- Radiolabeling and Stability: Document radioisotope labeling procedures (e.g. chelator chemistry for ^177Lu or ^68Ga) and provide stability data for the investigational product. Due to short half-lives, you must define shelf-life of prepared doses (often just a few hours) and specify time limits from prep to administration. MFDS will expect data on radiochemical purity over this period.
- Dose Calibration and Dosimetry: Clearly describe how patient doses are calibrated and measured. For therapeutic radionuclides, dosimetry calculations (e.g. organ dose estimates) should be part of the clinical protocol to guide safe dosing. Korean trial centers have strong nuclear medicine departments that can perform SPECT/PET-based dosimetry – plan to leverage these capabilities in Phase I/II to optimize dosing.
- Manufacturing Facilities: Identify the GMP-certified site where the radiopharmaceutical is made or compounded. Whether it’s an on-site hospital radiopharmacy or an external facility, MFDS will require a GMP compliance statement. Include the Annex 3 (radiopharmaceuticals) GMP certificate or reference. If using centralized manufacturing and shipping to sites, detail the packaging, cold chain, and time from production to injection.
- Quality & Safety Testing: Outline QC tests for each batch (radiochemical purity, endotoxin, sterility, pH, etc.). Because doses are used immediately, some tests may be post-release (e.g. sterility), but you should have a rapid sterility assurance plan (such as membrane filtration with results after administration). MFDS may ask how you’ll handle any out-of-spec results when the dose is already given – be prepared with a risk mitigation plan.
- CMC Variations: If your theranostic approach uses a diagnostic agent and a therapeutic agent (e.g. ^68Ga PET tracer and ^177Lu therapeutic), MFDS might want to see the relationship between the two. Ensure your IND covers the manufacturing and control of both the diagnostic and therapeutic components if they are part of the same trial.
Careful CMC planning not only satisfies regulators but also prevents disruptions during the Clinical Trial (e.g. batch failures or dosing delays). Engage local radiopharmacy experts early – Korea’s specialists can advise on meeting MFDS’s CMC expectations for radiopharmaceutical trials.
Radiation Safety Considerations in Clinical Trials
Radiation safety is a paramount concern in radiopharmaceutical Clinical Trials, and South Korea has a robust framework to protect patients, staff, and the public. Trial sponsors must incorporate radiation safety measures into both protocol design and site operations. Key considerations include:
- Regulatory oversight: In Korea, the Nuclear Safety and Security Commission (NSSC) oversees the use of radioactive materials, while MFDS focuses on drug safety. Hospitals conducting Clinical Trials with therapeutic radioisotopes must hold the necessary radioactive material licenses. Ensure each site is authorized to handle the specific isotope and activity level used in your trial. Most major Korean cancer centers and university hospitals have Nuclear Medicine departments with these licenses and standard operating procedures for radiotherapy isotopes.
- Site readiness and training: Verify that each trial site has a radiation safety program. This includes trained nuclear medicine physicians, medical physicists, and radiation safety officers. Staff should be trained in handling and administering radiopharmaceuticals, patient monitoring, and emergency procedures (like spill management). Patients may need to be isolated or have restricted contact for a period post-therapy (e.g. after high-dose I-131 or Lu-177 therapy) – sites must have appropriate facilities (shielded rooms or designated areas) for such Clinical Trials.
- Patient safety and monitoring: The protocol should detail how radiation exposure to patients is justified and minimized per ALARA principles. This involves dosimetry to ensure critical organs stay within safe exposure limits, routine blood counts to monitor for marrow suppression, and clear stopping rules for toxicity. Patients should receive radiation safety instructions – for example, guidance on limiting close contact with others for a defined time, proper hydration to flush out radioisotope, and what to do with bodily waste. Korean guidelines (mirroring IAEA recommendations) often require written instructions for patients after radioisotope therapy.
- Environmental safety and waste disposal: Plan for the safe disposal of radioactive waste (e.g. used syringes, IV lines, patient excreta). Trial sites in Korea have decay storage protocols where radioactive waste is stored until it decays to safe levels before disposal. The sponsor should verify that each site’s waste handling meets local regulations. During monitoring visits, Clinical Trial monitors may check that radiation survey meters, shielded storage, and waste logs are in place.
- Monitoring and compliance: It’s wise to include a radiation safety monitoring plan in the trial. This can involve periodic audits of radiation practices or requiring sites to report any incidents. South Korea’s sites generally have an excellent safety record, and the NSSC has technical standards in place to ensure ongoing compliance. Nonetheless, proactive oversight by the sponsor/CRO adds an extra layer of assurance.
By proactively addressing radiation safety in the trial design and site selection, sponsors can safeguard participants and staff while satisfying regulatory requirements. This approach also builds confidence among investigators and patients participating in radiotheranostic Clinical Trials.
Korea’s Role in Global Theranostic Development
South Korea is not only a place to run Clinical Trials – it’s an active contributor to the global rise of theranostics. The country’s commitment to innovation in radiopharmaceuticals is evident in both industry and policy actions. Local biotech companies are developing novel theranostic agents and rapidly advancing them through Clinical Trials. For instance, in 2025 FutureChem received MFDS approval to launch a Phase 3 trial of a prostate cancer radiotherapeutic (FC705), after showing promising Phase 2 results. Korean scientists and clinicians are also pioneering theranostic approaches in precision oncology, such as peptide receptor radionuclide therapy (PRRT) and PSMA-targeted radioligand therapy, often in collaboration with global partners.
The South Korean government actively supports this field. Recent MFDS regulatory reforms streamline Clinical Trials and approvals for innovative treatments. Agencies like MSIT (Ministry of Science and ICT) and KHIDI are investing in radiopharmaceutical R&D and infrastructure. As noted, domestic production of critical isotopes (Ac-225, I-131, etc.) is coming online to ensure self-sufficiency. Korea has also formed the Alpha Radiopharmaceutical Consortium with leading pharma companies to accelerate alpha-emitter drug development and propose supportive regulations. Such initiatives bolster Korea’s position as a theranostics powerhouse.
Globally, Korea is seen as a bridge between East and West in drug development. Data from Korean Clinical Trials are accepted by global regulators, allowing Korean trial results to be used in FDA or EMA submissions. This has encouraged multinational radiopharm sponsors to include Korea in pivotal trials for cutting-edge therapies. Moreover, Korea’s high recruitment speed and quality data (no FDA inspection failures to date) contribute to faster global approvals. The country often hosts key sessions in international nuclear medicine conferences, sharing insights from Korean trials and patients. All these factors underline Korea’s growing role in shaping theranostic clinical research worldwide.
Conclusion and Next Steps
Designing and operating a radiopharmaceutical Clinical Trial in South Korea requires meticulous planning across regulatory, logistical, and safety dimensions – but the payoff is significant. With MFDS’s efficient IND process, a skilled pool of investigators, and improving isotope infrastructure, Korea offers fertile ground to accelerate theranostic innovations. Sponsors who leverage Korea’s strengths – fast approvals, advanced imaging and dosimetry, experienced CRO support, and government incentives – can expect Clinical Trials that meet global quality standards while completing faster and at lower cost. In short, South Korea provides an ideal environment to “see it, treat it” in one seamless paradigm, fulfilling the promise of theranostics.
As the field of radiotheranostics continues to expand, South Korea will remain at the forefront, both as a trial destination and a source of innovation. Sponsors are encouraged to engage early with local experts to navigate MFDS requirements, secure isotope supplies, and ensure radiation safety from day one. A well-designed Korean Clinical Trial can not only advance your product to the next phase but also generate data credible worldwide – bringing life-saving theranostic treatments to patients faster.
FAQs: Radiopharmaceutical Clinical Trials in Korea
Q1: What is the MFDS process for approving radiopharmaceutical Clinical Trials in Korea?
A: The sponsor (or local representative) must submit an IND application to the MFDS with all required data (protocol, IB, CMC, etc.). The MFDS review takes ~30 working days. For radiopharmaceuticals, GMP documentation specific to radiodrugs is needed. Once MFDS grants IND approval and hospital IRBs approve the study, the Clinical Trial can start. Parallel IRB review often allows Korean trials to start faster than in other countries.
Q2: How are radioisotopes supplied for Clinical Trials in South Korea?
A: Many trial sites in Korea have on-site radiopharmacies to produce or prepare short-lived isotopes just in time. If importing isotopes, sponsors coordinate closely with licensed suppliers and customs for timely delivery. Korea is increasing domestic production of key isotopes like I-131 and Ac-225, which will improve supply stability. Multi-region trials often use Korea alongside other hubs (e.g. Australia) to ensure a reliable radioisotope supply chain.
Q3: What special CMC considerations apply to theranostic Clinical Trials?
A: CMC for theranostics must cover radioisotope handling and product stability. Sponsors should provide data on radiochemical purity, stability of the drug product (often only hours), and manufacturing under GMP for radiopharmaceuticals. Dose calibration and quality control testing (sterility, etc.) procedures must be detailed. MFDS will pay close attention to how you ensure each patient dose is safe and consistent given the short half-life and high potency of radiopharmaceuticals.
Q4: What are the radiation safety requirements for Clinical Trials with radiopharmaceuticals?
A: Trials must be conducted at sites with proper radioactive material licenses and trained nuclear medicine staff. Radiation safety protocols – covering patient isolation if needed, waste disposal, and staff protection – are mandatory. The hospital’s radiation safety committee typically reviews the study as well. Patients receive instructions to minimize radiation exposure to others after treatment (for example, hygiene and distancing guidelines post-therapy). Compliance with national radiation safety standards (overseen by NSSC) is required throughout the Clinical Trial.
Q5: Can data from Korean radiopharmaceutical Clinical Trials support global drug approvals?
A: Yes. Korea is ICH-GCP compliant and data from well-run Korean Clinical Trials are accepted by regulators like the FDA and EMA. Many global sponsors include Korean trial sites to gather data for international submissions. For instance, if a radiotherapeutic shows positive results in Korean Phase II trials, that data can be used in US or EU filings, provided the trial was robust. Korea’s quality and regulatory alignment make it an attractive part of a global development strategy for theranostic drugs.



