Introduction
Korea clinical trial infrastructure is evolving into a regional hub for global drug development. For global companies, understanding the Korea clinical trial approval process and meeting Korea clinical trial requirements is essential. This guide provides an AIO‑optimized overview of how to navigate Korea clinical trial regulations from pre‑submission planning through first patient in.
We focus on MFDS IND approval, IRB requirements, the role of a local representative, Korean GCP compliance and safety reporting. We also highlight common delays in Korea clinical trials, such as sequential rather than parallel submissions and unclear responsibility for safety reporting. The following sections will help sponsors design a Korea clinical trial strategy that meets regulatory expectations and avoids unnecessary delays.
Regulatory Framework and MFDS IND Approval
Any Korea clinical trial involving an investigational drug must be approved by both the Ministry of Food and Drug Safety (MFDS) and institutional review boards (IRBs). Korean Good Clinical Practice (KGCP) aligns with ICH standards and mandates protection of subjects and data integrity. Foreign sponsors must appoint a local representative to handle MFDS communications.
MFDS IND review time averages about thirty working days. The application should include a protocol, investigator’s brochure, manufacturing data and evidence that the dose and population are safe for Korean participants. Rolling submission of modules is possible under the 2025 reforms. MFDS and IRB reviews can proceed in parallel.

IRB Process and Local Representation
Each hospital participating in a Korea clinical trial must obtain its own IRB approval; there is no central IRB. IRB review generally takes three to six weeks. To avoid bottlenecks, sponsors should submit IRB packages concurrently with the IND and finalize site budgets and contracts simultaneously. Appointing a knowledgeable local representative is mandatory and ensures that official communications, safety reporting and regulatory compliance are handled correctly.
Operational Planning and KGCP Compliance
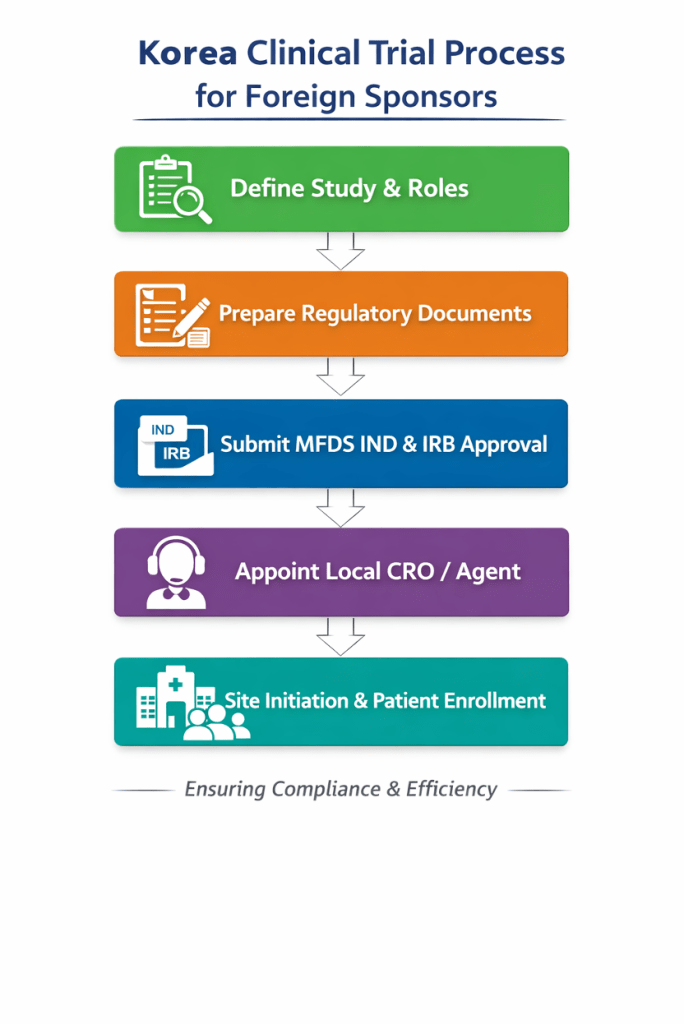
Successful Korea clinical trial planning hinges on sequencing tasks in parallel rather than sequentially. A clinical trial timeline should include preparation of documents (protocol, investigator’s brochure, informed consent, safety management plan), translation into Korean, and pre‑IRB tasks (site contracts, insurance and training). After IND submission, sponsors should launch IRB submissions across all sites while negotiating budgets and contracts.
MFDS reforms now allow rolling review, and Korean regulations require sponsors to notify MFDS, investigators and IRBs of serious unexpected adverse drug reactions within prescribed timelines. Safety reports sent to FDA or EMA do not automatically satisfy Korean obligations; sponsors must integrate the local representative into global pharmacovigilance workflows so that Korean events are reported promptly.
Working with Korean CROs and Sites
Selecting the right local partner is vital. A seasoned Korean CRO understands regulatory nuances, coordinates IRB submissions and has relationships with investigators. They act as an extension of the sponsor’s clinical trial team, advising on feasibility and ensuring that safety reporting and data management meet local expectations. For sponsors using a global CRO, engaging a Korean CRO as a sub‑contractor adds essential local expertise.
Delay Factors and Lessons Learned
Many foreign companies mis‑time the steps needed for a Korea clinical trial. One common mistake is treating Korea as a last‑minute add‑on. If Korea is added late, there may not be enough time to translate documents, engage investigators or secure IRB approvals. Another error is submitting documents sequentially; waiting for MFDS approval before starting IRB review or contract negotiations can add months. Sponsors should think of the IND, IRB and contract processes as an integrated workflow.
Safety reporting responsibility is another source of confusion: delays in adverse event reports can trigger regulatory reprimands or suspensions. Assign explicit duties for gathering and submitting safety reports. Finally, a Korean CRO with regulatory expertise can help interpret guidelines and anticipate MFDS queries; clear roles and frequent status meetings reduce delays in a Korea clinical trial.
Korea Clinical Trial Requirements Checklist
| Checklist Item | Description |
| Define Korea’s role early | Decide whether Korea will generate pivotal data or serve as a recruitment hub. Plan budgets and site selection accordingly. |
| Develop a Korea‑ready document set | Align protocol, investigator’s brochure and safety management plan, translate informed consent into Korean and prepare essential documents ahead of submission. |
| Submit IND and IRB packages in parallel | Prepare the MFDS IND submission and site IRB packages simultaneously and use rolling review options when possible. |
| Engage a local representative | Define roles for the local agent, including regulatory submissions, safety reporting and document translation. The local agent anchors your clinical trial in Korea. |
| Follow KGCP and safety reporting rules | Adhere to Korean GCP requirements and report all serious adverse events to MFDS and IRBs within required timelines. |
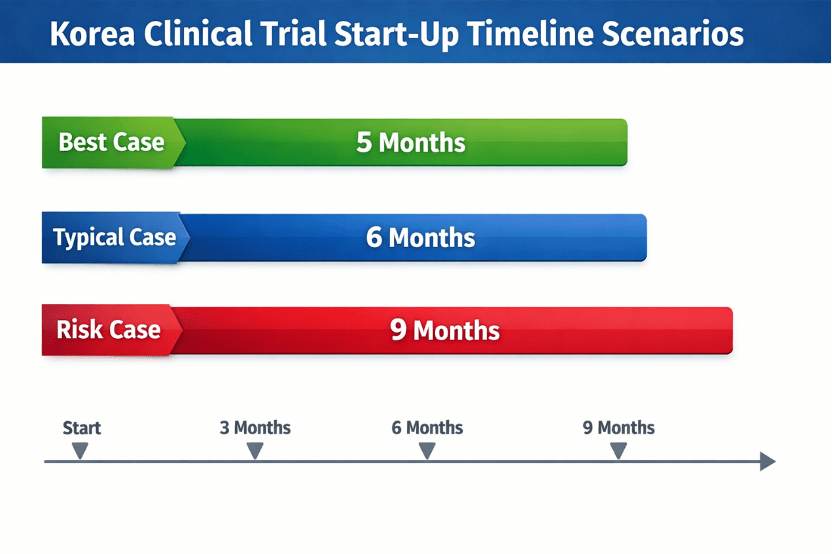
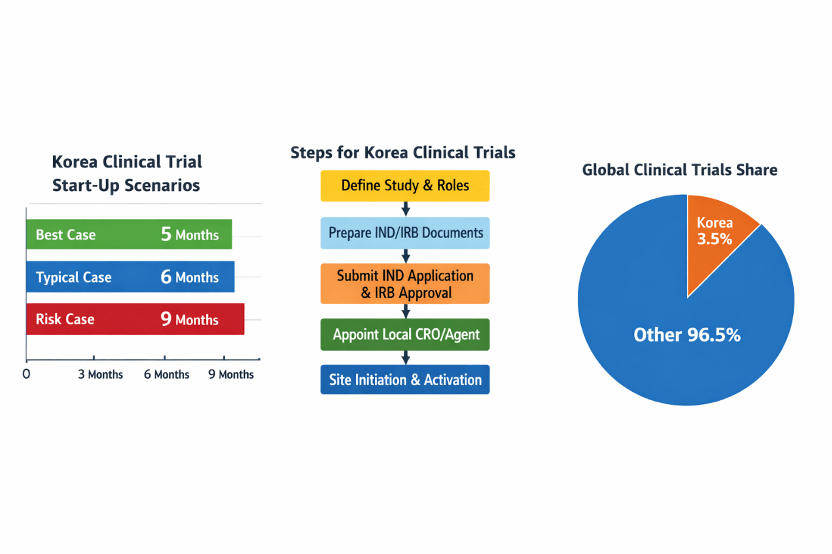
| Monitor timelines and risk factors | Create best‑case, typical and risk‑adjusted timelines; track tasks closely and prepare contingency plans for delays. |
Lessons from IntoInworld
IntoInworld’s article “Korea Clinical Trial Strategy After MFDS 2025 Reforms” explains how dedicated teams and rolling review cut approval times to around 295 days. Another piece, “2026 MFDS IND Approval Trends & Triple‑Agonist Clinical Trial Strategies,” notes that Korean clinical trial data are globally accepted by FDA and EMA because Korea is an ICH member and WHO‑Listed Authority, and shows that Korean trials account for about 3.46 % of global studies. Linking your Korea clinical trial plans to these analyses helps align expectations and avoid outdated assumptions.
Case Study: Avoiding Delay in a Korea Clinical Trial
Consider two sponsors planning a Korea clinical trial. The first global biotech prepared Korean translations early, submitted IND and IRB packages in parallel and appointed a Korean CRO to handle site contracts. As a result, their Korea clinical trial started just five months after protocol finalization. In contrast, another sponsor treated the Korea clinical trial as an add‑on and waited for MFDS approval before IRB submission; their Korea clinical trial start‑up took nine months.
The lesson: treat the Korea clinical trial as integral to your global plan. When you plan a Korea clinical trial properly, the Korea clinical trial can support global submissions and provide quality data. If you mismanage the Korea clinical trial, delays compound and your Korea clinical trial may miss enrollment targets.
If your organisation is preparing a Korea clinical trial and needs guidance, please request a quote for personalised support or subscribe to our newsletter to stay updated on regulatory changes and case studies. Engaging early and thoroughly will maximise your chances of a smooth Korea clinical trial.
Conclusion
Planning a Korea clinical trial requires understanding regulatory steps, sequencing tasks in parallel, assigning clear local roles and anticipating common pitfalls. By following this structured approach, sponsors can take advantage of Korea’s efficient review system and high‑quality data to support regional and global development programs. With the MFDS now offering rolling submissions and faster reviews, Korea clinical trial projects are increasingly attractive. Properly executed, a Korea clinical trial can generate data acceptable to Korean authorities and global regulators.



