Introduction
Korea’s clinical‑trial ecosystem is quickly becoming a global hub. In 2026, triple‑agonist clinical trials in Korea will dominate metabolic research. The Ministry of Food and Drug Safety’s (MFDS) IND approval trends and regulatory reforms are accelerating these studies, and data generated in Korea is officially recognized by the U.S. FDA and European EMA thanks to strict adherence to ICH‑GCP standards.
This article outlines how to leverage Korean data for global submissions in the WLA era, how to design and operate triple‑agonist trials, apply RBQM‑driven risk management, align with IRB and rolling review schedules, manage investigational product cold chains and collaborate with local CROs. It targets international pharma and biotech professionals seeking actionable insights.

Value of Clinical Trials in Korea & WLA Strategy
As an ICH member since 2016, Korea ensures global acceptability of its trial data. In 2024, Korean trials represented about 3.46 % of all global studies, and Seoul ranked as the world’s most active clinical‑trial city from 2017 to 2023. Large hospitals maintain robust electronic medical records and high patient compliance, producing reliable datasets. U.S. regulations (21 CFR 312.120) and European guidelines allow foreign studies meeting GCP to support approvals, enabling sponsors to design a single Korean trial for both domestic and foreign submissions.
Intoinworld’s article “Clinical Research Management in the WLA Era ” outlines five pillars for converting Korean data into global submission packages: data governance (define data elements and control versions), eCTD mapping (align Korean modules with U.S./EU electronic common technical documents), label concordance, inspection & quality bridge (translate local findings into foreign terminology and embed risk‑based quality evidence) and transfer package composition (prepare raw datasets, analysis code and label maps). Applying ICH E17 guidelines alongside this framework allows a single protocol to generate primary evidence acceptable across regions.

KRIs and RBQM Metrics (Table 1)
Triple‑agonist clinical trials require proactive monitoring using Risk‑Based Quality Management (RBQM). The table below presents key KRIs and their Quality Tolerance Limits (QTLs). Each indicator corresponds to a major risk; if a threshold is exceeded, a site‑level corrective and preventive action (CAPA) is required.
| Domain | Metric | Definition | Frequency | QTL (Signal Rule) |
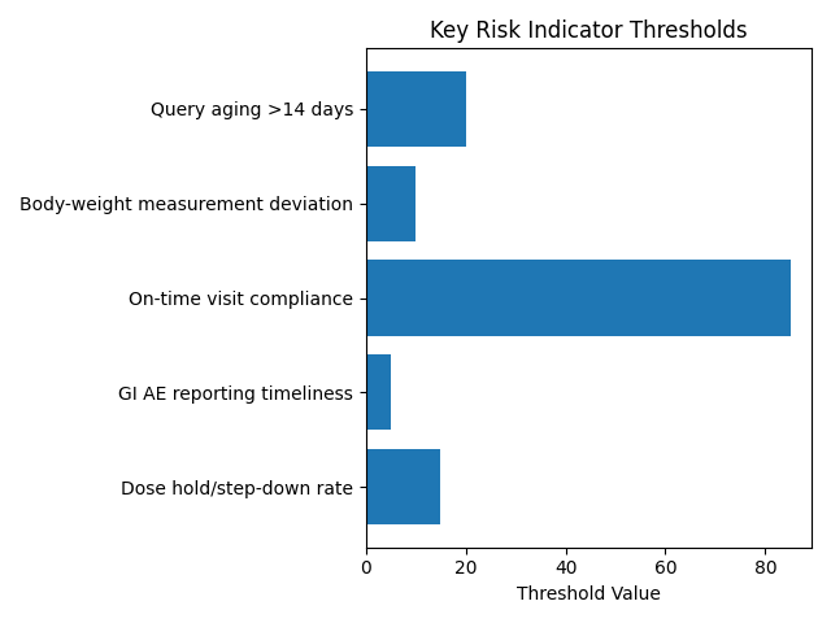
| Dosing | Dose hold/step‑down rate | Percentage of visits where dose escalation is held or decreased | Weekly | Warning if >15 % for three consecutive weeks |
| Safety | GI AE reporting timeliness | Median days from event occurrence to entry in the EDC | Weekly | Warning if median > 5 days for two consecutive weeks |
| Visits | On‑time visit compliance | Percentage of planned visits completed within ±3 days of the scheduled date | Weekly | Warning if <85 % for two consecutive weeks |
| Efficacy | Body‑weight measurement deviation | Percentage of weight measurements taken outside the allowed method or time window | Weekly | Warning if >10 % |
| Data | Query aging >14 days | Percentage of open data queries older than 14 days | Weekly | Warning if >20 % |
Each metric triggers an escalation when its threshold is breached.
Visualization of KRI Thresholds
The bar chart below visualizes the QTL values for each KRI. The dose‑hold/step‑down threshold is 15 %, GI AE reporting timeliness is 5 days, on‑time visit compliance is 85 %, body‑weight measurement deviation is 10 %, and query aging beyond 14 days is 20 %. These benchmarks form the basis for central monitoring to detect risk signals in real time.
Scientific Mechanism & Trial Design of Triple Agonists
Triple agonists simultaneously target GLP‑1, GIP and glucagon receptors to achieve superior glycemic control, weight loss and lipid metabolism improvements. Candidates like Eli Lilly’s retatrutide have shown >24 % weight loss over 48 weeks compared with ~6 % with single‑agonist semaglutide. The multi‑receptor mechanism requires meticulous management of gastrointestinal, gallbladder/pancreas and cardiovascular safety signals. Dose titration typically follows a four‑week stepwise escalation (low→mid→high dose) with clear hold/re‑challenge rules; moderate adverse events warrant holding the dose 2–4 weeks, symptomatic care and re‑escalation only when criteria are met.
Concomitant insulin or sulfonylurea should be down‑titrated before starting to mitigate hypoglycemia, and SMBG plus emergency contact plans must be in place. A hybrid visit schedule (onsite at weeks 0/4/8/12 and remote at weeks 2/6/10) balances participant convenience and data integrity. Locking the titration algorithm, SOPs and SAP versions ensures consistent treatment delivery across multiple centers.
Partnering with Intoinworld
To execute triple‑agonist trials successfully in Korea, sponsors need a partner fluent in local regulations and operations. Intoinworld is a Seoul‑based full‑service CRO with a strong hospital network and eTMF/central monitoring tools, offering WLA‑aligned trial design and evidence‑transfer strategies. Requesting a formal quote provides sponsors with tailored site selection, realistic timelines and risk‑management plans. To stay up‑to‑date on regulatory changes, we recommend subscribing to the Intoinworld newsletter for regular insights into Korea’s clinical‑trial environment.
IRB Review & Rolling Review Timeline
Starting a trial in Korea requires both site IRB approval and MFDS authorization. The IRB package should include a protocol with titration/hold tables, bilingual informed consent forms, culturally adapted recruitment materials, compensation plans, eConsent workflows and data‑privacy language. Most Korean IRBs meet monthly, so allow 4–8 weeks from submission to approval. The MFDS introduced rolling review in 2025, allowing modular dossier submissions that accelerate evaluation. Sponsors should prepare IRB documents and submit MFDS modules in parallel to compress overall timelines. Completing pre‑IRB tasks (site contracts, insurance, staff training) before submission prevents further delays. When timed well, IRB approval and rolling review align so FPI can proceed immediately after IND clearance.
IMP Cold‑Chain & Operational Management
Triple‑agonist products are often peptide injectables, so refrigeration and cold‑chain logistics are critical. IMP labels must include bilingual storage conditions, administration instructions and visit codes; randomization and blinding procedures should be documented and integrated into site training. Define step‑by‑step SOPs for temperature excursions (isolation, reporting, replacement kit dispatch) and establish a RACI matrix delineating responsibilities between sponsor, CRO and sites. Using a central depot near Incheon Airport and shipping only 1–2 weeks of stock to sites simplifies temperature monitoring and inventory control.
Recruitment & Retention Strategies
While Korea’s obesity rate is lower than in the West, the concentration of metabolic clinics around Seoul allows adequate recruitment. EMR pre‑screening identifies candidates meeting BMI and glycemic criteria, and integrating lifestyle counseling into clinic workflows supports enrollment. To minimize attrition in long trials, combine hybrid monitoring, weekly calls, family education and travel reimbursement. Emphasizing engagement and convenience can sustain visit‑completion rates above 90 %.
Additional Resources
Further details and case studies are available in the Intoinworld Clinical Trial Information section, which covers topics such as WLA‑era clinical research management, CDx co‑development strategies and ADC trial trends. Additional datasets and the Excel file referenced here will be provided separately for download and analysis.

Summary
The MFDS IND landscape is evolving rapidly, offering new opportunities for overseas sponsors planning triple‑agonist clinical trials in Korea in 2026. Applying the WLA framework and ICH E17 allows a single Korean study to support multiple regulatory submissions. Success hinges on RBQM‑driven KRI monitoring, precise dose titration and safety management, efficient IRB/rolling review processes, cold‑chain logistics, patient‑retention strategies and partnership with an experienced local CRO. We hope this guide provides practical value to pharma and biotech professionals leveraging Korea as a clinical‑trial hub.



