Why korea strategic clinical trial environment matters
Ranking the best‑selling drugs of 2025 is more than a corporate scoreboard. The names at the top—Keytruda, Ozempic, Mounjaro, Dupixent and Skyrizi—show where R&D budgets, clinical trial pipelines and regulatory scrutiny are converging. Yet none of these blockbusters would succeed without a stable environment for medical trial execution. In early 2025 the Korean Ministry of Food and Drug Safety (MFDS) overhauled its new‑drug review process: it created dedicated review teams, expanded sponsor consultations, introduced rolling submission of CTD modules and conducted GMP evaluations in parallel, cutting approval timelines from about 420 days to 295 days . These reforms position Korea not as a peripheral Asian country but as a strategic hub for global development.

MFDS 2025 Reforms:
Why Korea is Becoming a Strategic Hub
Summary of the Top 5 Drugs
| Drug | Mechanism & key indications | 2025 projected sales (USD bn) |
| Keytruda (pembrolizumab) | PD‑1 checkpoint inhibitor; numerous cancers including non‑small‑cell lung cancer, melanoma, head & neck | 30 |
| Ozempic/Wegovy (semaglutide) | GLP‑1 receptor agonist; type 2 diabetes and obesity | 20 |
| Mounjaro (tirzepatide) | Dual GIP/GLP‑1 agonist; diabetes and obesity with greater weight reduction | 19 |
| Dupixent (dupilumab) | IL‑4/IL‑13 antibody; atopic dermatitis, asthma, chronic rhinosinusitis and other Type 2 inflammatory diseases | 17 |
| Skyrizi (risankizumab) | IL‑23 antibody; plaque psoriasis, psoriatic arthritis, Crohn’s disease | 15 |
These drugs share features that drive blockbuster revenue: they treat chronic or life‑threatening conditions, serve multiple indications and require long‑term therapy. Such complexity demands robust clinical research management to design protocols that satisfy regulators across regions.
Chart: Projected Sales of the Top 5 Drugs
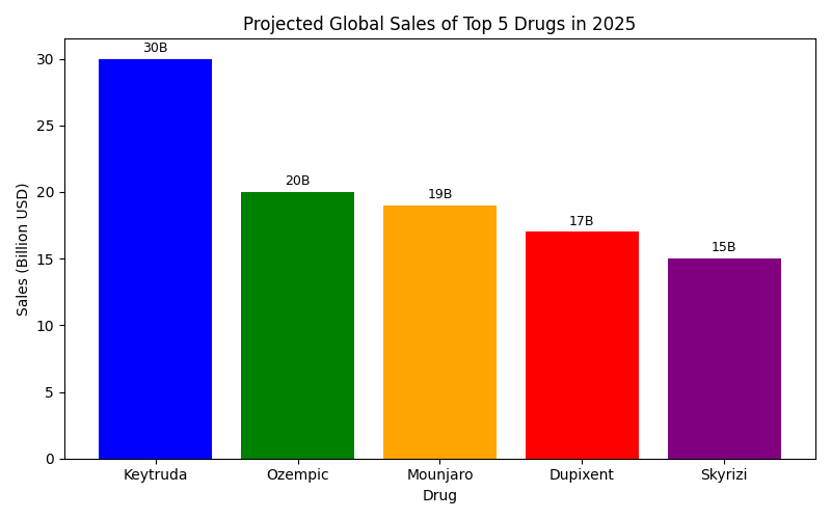
The bar chart illustrates the relative scale of each drug’s forecast revenue. Keytruda leads by a wide margin due to its use across numerous cancers, while GLP‑1‑based therapies like Ozempic and Mounjaro show explosive growth.
What These Products Tell Us About Future Clinical Trials
- Oncology and immunotherapy – Keytruda’s success foreshadows more basket and umbrella trials testing one mechanism across multiple cancers. Combinations and immune‑related adverse‑event monitoring will become integral to trial design.
- Metabolic and obesity medicine – Ozempic and Mounjaro collapse the line between glycaemic control and weight management. Expect large cardiovascular‑outcome trials and real‑world evidence studies linking weight loss to comorbidities such as fatty liver disease and sleep apnoea.
- Chronic inflammatory and autoimmune diseases – Dupixent and Skyrizi show how targeting specific cytokines can unlock multiple indications. Composite endpoints, patient‑reported outcomes and long‑term safety follow‑up will grow in importance.
These trends require an agile clinical research organization capable of running integrated programmes across disease areas. Collaboration with a clinical research contract research organization ensures consistent data packages for MFDS, FDA and EMA submissions.
MFDS 2025 Reforms and the New Korean Landscape
The 2025 MFDS reforms fundamentally restructured drug review. A single lead reviewer is replaced by a 10–15‑member team covering clinical, non‑clinical, CMC and regulatory disciplines. Official meetings have expanded from about three to ten or more, allowing real‑time discussion of supplementary data. Rolling review enables sponsors to submit CMC and non‑clinical modules early, while GMP inspections occur in parallel. Korea is listed as a WHO Listed Authority (WLA) and has long been an ICH member; about 3.46 % of sponsor‑led global trials are already conducted in Korea. Because trials follow ICH‑GCP, data generated in Korea can support FDA and EMA approvals when the study is well designed.
Chart: MFDS Approval Timeline Reduction
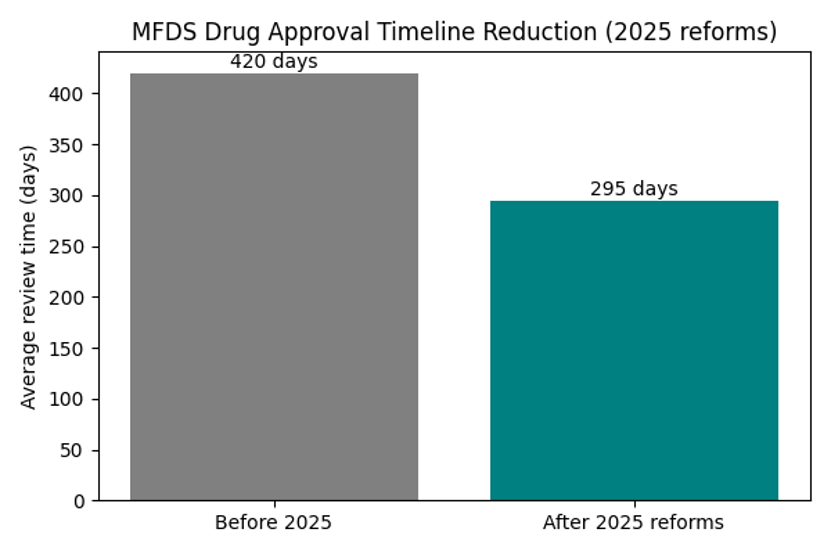
The chart highlights how rolling review and parallel GMP evaluation shrink the average approval time. This acceleration means faster patient access and greater capital efficiency.
Pie Chart: Share of Sponsor‑led Trials in Korea
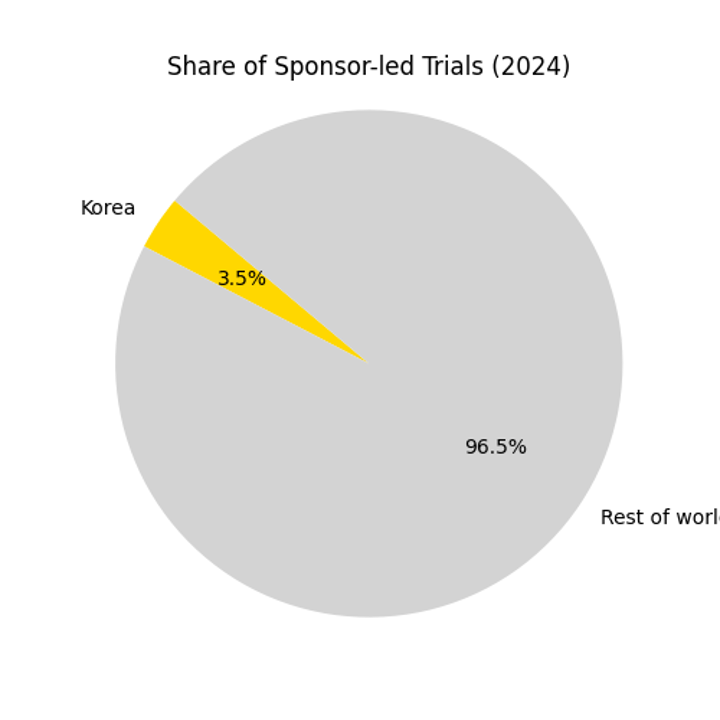
Though Korean trials currently account for only about 3.46 % of the global total, the WLA listing and regulatory reforms create ample room for growth. Seoul consistently ranks among the top cities for clinical research.
The Value of Korean CROs
Executing a successful korea clinical trial requires local expertise. A Korean clinical research organization can translate MFDS requirements into practical protocols, stress‑test global designs against local standard of care and anticipate operational bottlenecks. Intoinworld goes further by implementing five pillars for globally submission‑ready evidence: data governance, eCTD mapping, label concordance, inspection and quality bridge, and transfer‑package composition. Working with an experienced partner enables sponsors to build reusable site networks and data cultures rather than one‑off projects.
Why choose Intoinworld?
Intoinworld goes beyond generic CRO services. The company has built experience across the very disease clusters represented by the top‑selling drugs—oncology, metabolic disorders and chronic inflammatory diseases—so it understands recruitment challenges and endpoint expectations. Through close collaboration with local investigators, it devises realistic enrolment strategies, secures clean, inspection‑ready data and designs protocols that anticipate long‑term extension studies. By operationalising the five pillars of data governance, eCTD mapping, label concordance, inspection & quality bridge and transfer‑package composition, Intoinworld ensures Korean datasets can be easily validated by FDA and EMA reviewers. For these reasons, Intoinworld is not just a service provider but a strategic partner for entering the Korean market.
Considering your first or next korea clinical trial? Reach out via Intoinworld’s quotation request form to discuss your project. For ongoing insights into MFDS guidance and clinical research trends, sign up for their newsletter. Collaborating with a seasoned Korean CRO can transform local studies into globally accepted evidence.



