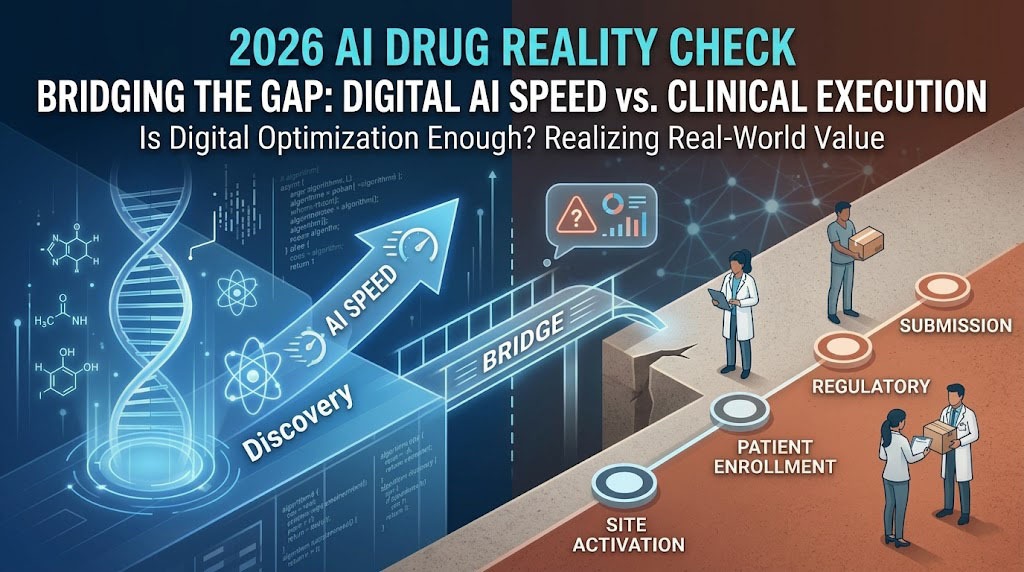
The year 2026 marks a pivotal era where AI-generated drug candidates are delivering their first wave of pivotal clinical readouts. While AI has successfully compressed the “Discovery” phase from years to months, a critical bottleneck remains: Clinical Execution. Many Sponsors find that the “AI speed” gained in the lab is lost during the slow, traditional processes of patient recruitment and regulatory filing.
As a leading CRO in South Korea with over a decade of expertise, Intoinworld provides the strategic bridge to ensure your AI-driven assets maintain their momentum through to market approval.
1. The 2026 Global Landscape: From “In-Silico” to “In-Vivo” Proof
By Q2 2026, over 170 AI-native pipelines are in clinical stages. The industry has moved beyond the hype, focusing now on Clinical Proof-of-Concept (PoC). Recent data shows that while AI-designed molecules have a higher Phase I success rate (approx. 85%), Phase II remains the ultimate “valley of death” where human biological complexity meets digital prediction.
2. Intoinworld’s Core Insights: Synchronizing the AI Advantage
“Digital Twin” Feasibility for Hyper-Precise Recruitment
AI drugs often target niche biomarkers, making traditional recruitment methods obsolete.
- The Intoinworld Solution: We utilize South Korea’s centralized Electronic Medical Record (EMR) systems to perform “Virtual Feasibility Scans.” Before your protocol is even finalized, we identify the exact patient clusters across our Tier-1 hospital network. This transforms recruitment from a passive wait into an active, data-driven strike, cutting timelines by up to 50%.
Navigating ICH E6(R3) with Predictive Quality Control
The 2026 implementation of ICH E6(R3) places unprecedented emphasis on Data Integrity.
- The Intoinworld Solution: Our proprietary “Predictive QC” system operates at the site level. As data is entered, it is cross-referenced against AI-modeled expectations. If an anomaly occurs, our system triggers an immediate corrective action. For global Sponsors, this means a “clean” data package that withstands the scrutiny of FDA or EMA inspectors, drastically reducing the risk of Refusal to File (RTF).
3. Strategy Comparison: Traditional vs. 2026 AI-Driven R&D
| Stage | Traditional R&D (10-12 Years) | 2026 AI-Driven R&D (5-7 Years) | Intoinworld Acceleration Point |
| Discovery | 3-5 Years | 1-2 Years | Regulatory consulting to ensure “approvability” of AI leads. |
| Pre-clinical | 1-2 Years | 0.5-1 Year | [Speed] Translating AI simulations into MFDS/FDA-ready protocols. |
| Phase I/II | 3-5 Years | 2-3 Years | [Core] Hyper-fast recruitment via Korea’s high-density hospital net. |
| CSR / Filing | 6-12 Months | 2-3 Months | [Speed] AI-automated SAS programming to cut reporting time by 40%. |
Q&A: Strategic Insights for Global Sponsors
Q1: Why do AI-designed drugs still face high Phase II failure rates? A1: AI excels at optimizing binding affinity, but predicting complex human metabolism remains a challenge. Intoinworld mitigates this by integrating local Asian PK/PD data early, identifying safety signals that digital models might miss.
Q2: How does Intoinworld ensure data security for our proprietary algorithms? A2: We employ an ISO-certified, encrypted environment. Your AI models remain your intellectual property; we focus on the clinical data integrity required for global regulatory submission.
Q3: Can South Korean clinical data be used for concurrent FDA/EMA filings? A3: Absolutely. South Korea is a member of ICH, and our data is highly regarded for its quality and consistency. Intoinworld specializes in bridging Korea-specific data into global eCTD packages.
Q4: How do you handle recruitment for ultra-rare biomarkers identified by AI? A4: Through our direct partnerships with Korea’s top medical centers, we can access specialized genomic databases (K-Biobank) to locate rare patient profiles that are difficult to find in Western markets.
Q5: What is the cost-benefit of running AI drug trials in Korea with Intoinworld?
A5: You gain “First-World” clinical quality and high-density data at a significantly lower cost-per-patient compared to the US or EU, while maintaining the speed required by AI-driven timelines.