Why “pan-RAS inhibitor payload ADC” is suddenly being discussed so much
Over the last 1–2 months, one phrase has been showing up more often in oncology R&D conversations: not “another ADC,” but <b>the payload itself is changing</b>. Since AACR 2026, the center of gravity has shifted further from “which antigen do we target?” to “what mechanism do we deliver, how do we deliver it, and how controllable is that delivery?”
In that context, a pan-RAS inhibitor payload is particularly symbolic. RAS has always been a pillar of oncology biology and drug development, but it has also been persistently difficult—especially when you talk about broader pathway inhibition. The broader the inhibition concept becomes, the more sensitive the tolerability and safety window can be. That’s why recent discussion is moving beyond “let’s make a new inhibitor” toward “can we redesign <b>how</b> inhibition is delivered so the therapeutic window is rebuilt?” One approach is to take a mechanism-type small molecule and turn it into an ADC payload so it can be delivered more selectively to tumor cells.
From “target competition” to “payload competition”
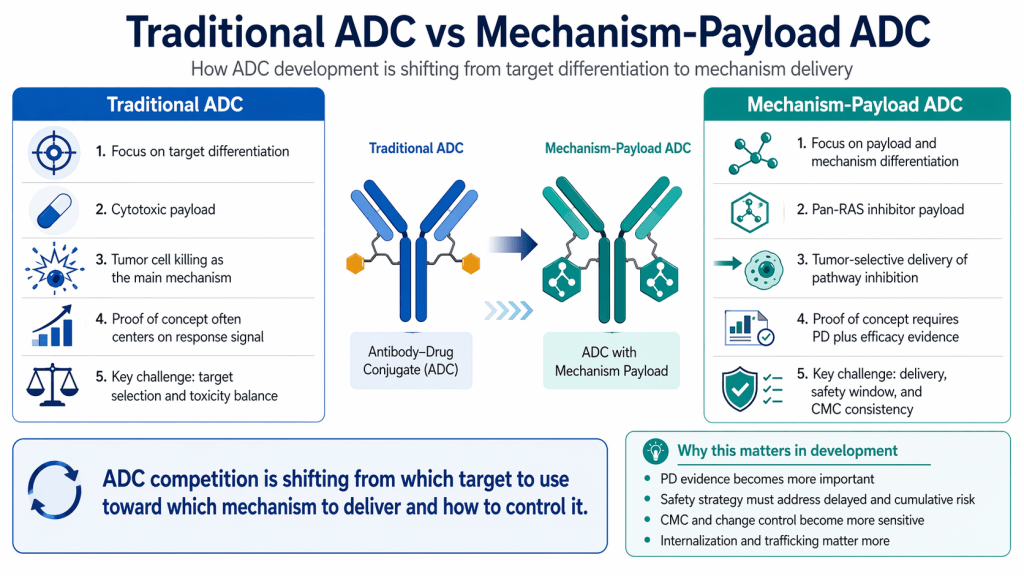
Traditional ADC logic is easy to summarize: attach a potent cytotoxic payload, bind to tumor cells, internalize, release payload, kill cells. When clinical responses appear, the story can be persuasive and intuitive.
A pathway mechanism payload (like a pan-RAS inhibitor) changes the logic. For pathway inhibition, success often depends on exposure time, tissue distribution, metabolism, and the impact on normal tissues. Converting that mechanism into a payload is not simply “loading toxicity.” It’s an attempt to <b>re-tune where and how strongly the mechanism operates</b> through delivery design.
That’s also why this approach cannot rely on binding alone. It needs consistent internalization, predictable intracellular processing, and reliable release behavior—because those steps determine whether the payload reaches effective intracellular exposure in the right cells. This is why designs like <b>biparatopic</b> binding often appear in the same narrative. In plain terms: as payloads become more complex, the delivery performance of the antibody platform becomes more central.
Why mechanism-payload ADCs feel harder in real development
It’s tempting to treat this as a “cool concept that should work,” but development rarely becomes easier automatically. Instead, it creates new practical questions—questions that are not glamorous in papers, but that often decide whether a program advances smoothly or stalls.
In mechanism-payload ADCs, clinical design, safety strategy, CMC decisions, and data operations don’t sit in separate lanes. The mechanism can make efficacy and safety move together on the same axis, manufacturing or comparability changes can become interpretation risk, and data alignment issues can directly weaken the credibility of the evidence. “Complexity” here is not an abstract word—it means the <b>difficulty of building and defending an evidence narrative</b> increases.
Four development shifts sponsors should plan for
Proof of concept cannot be “ORR only”: PD becomes the backbone
With cytotoxic payload ADCs, early PoC can sometimes lean heavily on visible response signals. With a pan-RAS inhibitor payload, the questions come faster: “Was this truly RAS pathway inhibition?” “Was inhibition durable?” “Who benefits most and why?” Those questions are even more central in global review and partnering discussions.
Practically, PD should not be treated as a nice-to-have appendix. For mechanism-payload ADCs, PD should be part of the program’s explanation structure. That means defining what to measure, when to measure it (not just one timepoint), and how to connect PD changes to clinical outcomes (including responder vs non-responder patterns). When that backbone is strong, limited response signals can still be interpretable, and strong responses become more defensible as reproducible biology rather than luck.
Safety is not “automatically better”: new toxicity patterns can emerge
“Tumor-selective delivery” is a powerful promise, but it is not a guarantee. Safety may still be shaped by normal tissue target expression, payload release and metabolism, and the practical extent of bystander effects.
So the safety strategy should not stop at “typical ADC toxicities.” It should assume that <b>delayed</b> and <b>cumulative</b> risks may matter and design monitoring accordingly. The practical goal is not “monitor more,” but “monitor smarter”: define which signals matter, how often they are reviewed, and what escalation rules apply. If those rules are defined early, safety data accumulates into a structured narrative rather than a messy set of events.
CMC and change control can’t be postponed: payload changes may become program changes
Mechanism-type payloads behave more like “the drug itself.” Small changes in linker, payload, or process can invite stronger questions about impact on efficacy and safety. Many teams try to move fast by postponing CMC rigor, but this category can punish that approach later.
The practical shift is that batch consistency, the rationale for release specifications, and the bridging logic for inevitable changes should be thought through earlier. This is not only for regulators. It also matters for BD because partners are assessing whether the platform can be scaled to multiple assets. CMC and change control are not just “factory issues”—they can become <b>deal value</b> and <b>development risk</b> issues.
Internalization and trafficking variability becomes more consequential
For these programs, the extent and consistency of intracellular delivery and processing can directly shape outcomes. High target expression alone may not be sufficient. Differences in cellular processing capacity, tumor microenvironment, and how closely real-world biology matches preclinical delivery assumptions can all matter.
As a result, clinical strategy may increasingly revolve around a sharper question: in which patient populations does delivery occur reliably enough for the mechanism to be consistently demonstrated? If that is answered well, PoC probability improves and data looks cleaner. If not, the program can appear unstable across sites or subgroups, and interpretation costs rise.
Practical insight for China-based developers planning overseas trials
For China-based teams, mechanism-payload ADCs can be highly attractive: differentiation is clear and partnering stories are easier to tell. But they can also be categorized as higher-risk assets, which means overseas partners and investors tend to demand a <b>system that explains and controls the risk</b>. Overseas trials should not be treated as “the same study in another country.” They should be treated as building a <b>global-readable evidence package</b>.
In practice, overseas PoC becomes much smoother when these six elements are designed upfront:
First, a PD/mechanism evidence plan (samples, timing, assays).
Second, a safety monitoring structure built around delayed and cumulative risk assumptions.
Third, CMC consistency and basic change control principles with bridging logic.
Fourth, central lab readiness and sample traceability.
Fifth, an EDC–Safety–PK/PD reconciliation rhythm so the evidence stays coherent.
Sixth, multi-criteria go/no-go thresholds (not ORR alone).
These are not about producing more documents. They are about building an operating system that stays stable as the program scales—so the next program runs faster, and the cost of answering partner questions goes down.
Conclusion: in 2026, payload mechanism is becoming the key development language
Mechanism-payload ADCs—illustrated by pan-RAS inhibitor payload concepts—signal that ADCs are expanding beyond cytotoxic carriers toward <b>mechanism delivery platforms</b>. That shift changes the shape of PoC evidence, the safety narrative, CMC and change control expectations, and the data governance needed to keep the story coherent. Over time, the core question becomes less “what target is it?” and more “what mechanism are you delivering, and how do you prove and control it?”
Intoinworld is a Korea-based CRO with over 10 years of local execution experience across Phase I–IV and post-marketing studies. If you are planning overseas PoC or global-ready execution for novel modalities (including ADCs), we can support IND/IRB readiness, site operations, data management, and safety operations end-to-end. Please use the Quotation button on this page to request a quote.



