Introduction
Clinical trials rely on two complementary roles: the CRA and the CRC. A CRA works for a sponsor or contract research organization and travels between sites to monitor trial progress and regulatory compliance, while a CRC works at the hospital, recruiting participants, coordinating schedules and managing study documentation. This article compares their duties and explains how they work together in Korea’s advanced clinical trial infrastructure.
Role Comparison
CRAs and CRCs differ in affiliation and scope. A CRA, affiliated with a sponsor or CRO, visits multiple sites to check drug storage, verify patient consent and data accuracy, and prepare reports for sponsors and regulators. A CRC, based in the hospital, explains the study to participants, obtains consent, schedules treatments and tests, and enters data. The bar chart above illustrates the relative emphasis of their tasks.
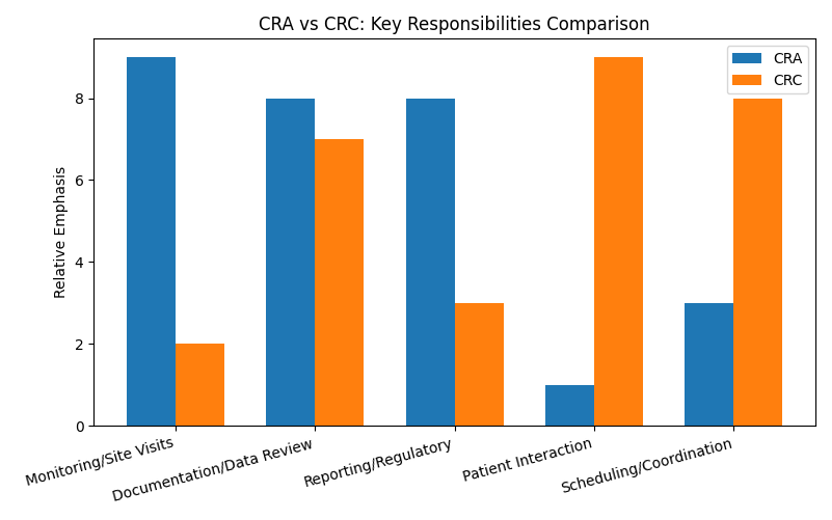
As shown, CRAs devote more time to monitoring, quality control and regulatory interaction, whereas CRCs focus on patient care and logistical coordination. CRAs ensure that trial procedures align with the protocol and report deviations, while CRCs interact with participants and prepare documentation to support the CRA’s review.
CRA Responsibilities and Skills
CRAs monitor the overall conduct of a trial. Key duties include site visits, checking investigational product storage, verifying patient enrollment and consent, reviewing data for completeness, ensuring compliance with GCP and protocols, and writing monitoring reports. CRAs typically hold degrees in pharmacy, life sciences or nursing and complete Good Clinical Practice training. They require regulatory knowledge, data review skills and strong communication.
CRC Responsibilities and Skills
CRCs manage on-site operations. Their duties include recruiting participants, explaining the study and obtaining informed consent, managing drug administration and test schedules, preparing documentation for CRA visits, coordinating with physicians and nurses, and reporting adverse events. CRCs often have healthcare degrees such as nursing or clinical laboratory science and need skills in patient interaction, scheduling and teamwork.
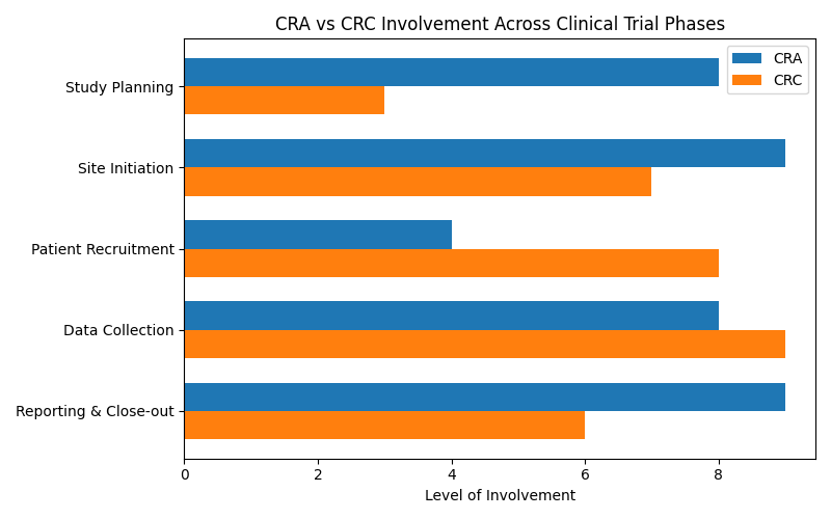
Roles Across Trial Phases
Clinical trials proceed through study design, regulatory approval, conduct, data management and close-out. CRAs play a major role in the design and approval stages, ensuring that protocols and documentation meet regulatory requirements. During the conduct and data phases, CRCs handle participant management and scheduling, while CRAs review data quality and compliance. The following chart visualizes their relative involvement at each stage.
The Korean Context
Korea has emerged as a global clinical trial hub, offering advanced infrastructure and strict regulatory oversight. Professional CRAs continuously monitor sites and internal quality systems evaluate data integrity; all trial personnel receive regular GCP training. CRCs, often trained as research nurses, support rapid patient recruitment and efficient site operations. This environment ensures that CRA feedback is quickly implemented and that trials meet international quality standards. Korea’s clinical trial costs are 30–40% lower than those in the United States while maintaining comparable quality and faster enrollment, which reduces monitoring expenses.
For more information on trial procedures and strategies, visit Intoinworld’s Clinical Trial Information page.
Conclusion
CRAs and CRCs are two pillars of clinical trial execution. The CRA monitors protocol adherence and data quality, while the CRC manages participant interactions and day‑to‑day logistics. Their collaboration is essential for efficient and safe trials. Intoinworld provides global‑standard services through an experienced network of CRAs and CRCs. For assistance or cost estimates, visit our Request a Quote page, and subscribe to our Newsletter for the latest insights.
FAQ
Q1: What is the main difference between a CRA and a CRC?
A1: A CRA works for a sponsor or CRO and travels between trial sites to monitor compliance and report findings, while a CRC is based at a hospital and handles patient recruitment, scheduling and data entry.
Q2: How do CRAs and CRCs collaborate?
A2: CRAs review documents and data prepared by CRCs and provide feedback; CRCs adjust site operations according to this feedback and continue managing patients. Together they ensure trial quality and participant safety.
Q3: What qualifications are needed to become a CRA or CRC?
A3: CRAs typically hold degrees in pharmacy, life sciences or nursing and must understand regulations and data analysis. CRCs generally have nursing or clinical lab backgrounds and need strong patient interaction, scheduling and teamwork skills.

