Clinical trials are regarded as the “Cornerstone of drug development”, representing an essential phase in the process. However, they demand substantial time and financial investment, with a success rate of less than 10%. Furthermore, amid the rapidly evolving regulatory environment and various unforeseen challenges, pharmaceutical and biotechnology companies actively collaborate with Clinical Research Organizations (CROs) to advance clinical expertise and operational efficiency.
South Korea is rapidly emerging as a global hub for clinical trials, attracting considerable interest from international pharmaceutical companies due to its robust patient pool and cutting-edge infrastructure. Consequently, choosing a trustworthy Clinical Research Organization (CRO) is vital for ensuring project success. An improper choice may not only lead to significant time and cost inefficiencies but also adversely affect the quality and outcomes of the research, thereby underscoring the importance of a strategic approach in the CRO selection process.
Korea CRO Market: Current Status and Advantages
Over the past two decades, South Korea’s clinical trial market has experienced remarkable growth. By 2024, the country is conducting approximately 3.46% of all global sponsor-driven clinical trials, ranking it sixth worldwide, behind the United States, China, Australia, Spain, and Germany. Seoul consistently ranked as the top city for clinical trials globally from 2017 to 2023, maintaining its position as the second-largest hub in 2024. This remarkable progress is largely attributed to strong government support and the development of advanced hospital infrastructure.
More than 200 medical institutions across the country have been designated as clinical trial sites, enabling access to a large and diverse patient pool, particularly from major hospitals. In addition, the country has established high-quality data management systems based on Electronic Medical Records (EMR) and a stringent quality control system aligned with ICH-GCP standards. This commitment to quality has enabled South Korea to maintain high clinical trial standards, with no major issues reported during FDA inspections over a prolonged period.
Amid the rapid development of the clinical trial sector, Korea CRO industry has also experienced significant expansion. Today, approximately 65 CRO companies operate in Korea. Over the past few years, industry has demonstrated both quantitative and qualitative growth, with the workforce increasing by 47.4% over the past 3 years and revenue rising by 77.7% in the last 5 years.
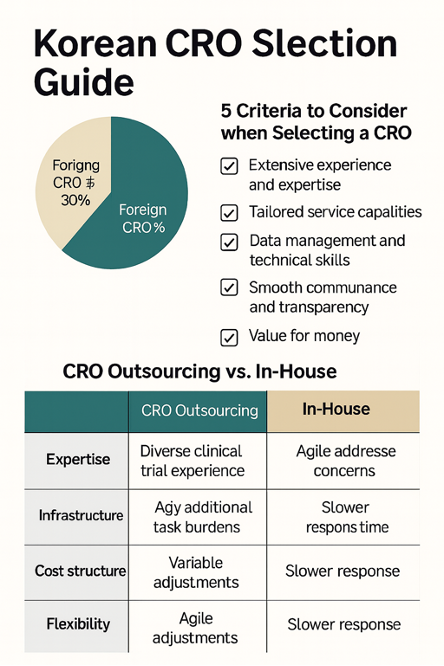
In the Korea market, multinational global CROs and domestic CROs coexist. Approximately 70% of the domestic CRO market is held by large global CRO’s, while the remaining 30% is shared among 20 to 30 domestic companies. Leading global CROs leverage their substantial capital and extensive global networks to secure projects within Korea, whereas domestic CROs expand their market presence by emphasizing local expertise and cost competitiveness.
When conducting clinical trials in Korea, costs can be 30 to 40% lower than in the United States while maintaining comparable research quality. Furthermore, the regulatory approval process by the Ministry of Food and Drug Safety (MFDS) is typically completed within an average of 8 weeks, allowing for faster regulatory approval than in Japan (10–12 weeks) or China (12–16 weeks).
Korea CROs have demonstrated strong performance in global clinical trials by combining robust patient recruitment capabilities, rigorous quality management, and cost-effectiveness. For instance, in a Phase 3 clinical trial for diabetes medication conducted by an international pharmaceutical company, partnering with a Korea CRO led to a 35% reduction in costs compared to initial estimates and shortened the trial period by 6 months. These results highlight the significant business value that can be achieved through the selection of the right CRO partner.
CRO Outsourcing VS. In-House Clinical Trials
Although companies can conduct clinical trials using solely their internal resources, an increasing number of organizations are choosing to outsource clinical trials. This is particularly common among small and mid-sized pharmaceutical and biotech companies, which often face challenges in maintaining specialized personnel and infrastructure internally. Furthermore, limited experience in regulatory compliance and data management increases the risk of costly trial-and-error processes. Collaborating with experienced CROs helps reduce operational risks and shortens development timelines. The table below outlines the key differences between CRO utilization and in-house clinical trial execution.
| Category | Utilizing CRO (Outsourcing) | In-house Clinical Trial Execution |
| Expertise & Experience | Possesses professional personnel across diverse therapeutic areas with extensive clinical trial experience. Comply with the latest regulations and international standards (e.g., ICH-GCP). Demonstrates strong capability in anticipating and addressing potential issues in advance. | Limited experience outside specific therapeutic areas. The internal team requires time to build comprehensive expertise, leading to a learning curve. Difficulty in keeping up with the latest regulatory trends. |
| Infrastructure & System | Operates specialized platforms & SOPs such as data management systems (EDC), safety monitoring, and statistical analysis. Quality Assurance (QA) team established and operated. | Lack of dedicated systems may result in additional time and costs for setting up. Existing staff may need to take on additional responsibilities, increasing the potential for errors. |
| Cost Efficiency | Variable cost structure depending on project duration. Professional outsourcing enables cost optimization and potential long-term cost reduction. | Fixed and ongoing personnel costs due to direct employment. Additional costs arise for implementation. Inefficient for small-scale or irregular projects. |
| Flexibility & Timeliness | Experienced in managing multiple projects, enabling rapid progress. Flexible in resource and manpower allocation to meet timelines and respond efficiently to urgent issues. | Limited internal resources may cause delays when workload increases. Response to unexpected issues may be delayed, making it difficult to ensure schedule stability. |
| Regulatory compliance capability | Expert in MFDS, FDA, and EMA regulations, with systematic management of the latest regulatory updates | Lack of a regulatory team can cause submission delays and repeated revisions; limited knowledge of global clinical regulations may lead to trial-and-error approvals |
Although some major pharmaceutical companies operate their own clinical teams, most sponsors usually contract specialized CROs to conduct clinical trials. In countries like Korea, where clinical trial infrastructure is well-established, partnering with a CRO with local expertise provides advantages in both speed and quality of development.
5 Key Criteria for Selecting a Korea CRO
The selection of an effective clinical research organization (CRO) requires a comprehensive evaluation of multiple factors. 5 core criteria should be given particular attention: experience and expertise, capability for customized services, technical capabilities and data management, communication and transparency, and value for cost. The following outlines the specific aspects to consider for each of these criteria.
1. Extensive Experience and Expertise
A CRO’s experience and expertise in clinical trials are the top factors to consider. It is essential to verify whether the candidate CRO has sufficient experience in the sponsor’s therapeutic area. For instance, review whether they have a proven track record in managing clinical trials for the relevant indication, covering study design, patient recruitment, and data analysis. Equally important is assessing whether the CRO’s team is well-versed in international standards such as ICH-GCP and familiar with MFDS approval procedures. A partner with demonstrated ability to manage complex trials and up-to-date regulatory knowledge can be regarded as a reliable choice.
2. Customized Service Capability
The success of a clinical trial often depends on a CRO’s ability to provide flexible, project-specific services. It is important to ensure the CRO has experience delivering customized solutions rather than a standardized approach. For example, it is important to review whether the CRO has experience flexibly adapting trial designs to accommodate budget or timeline constraints, and whether they have demonstrated prompt and effective responses to unpredicted issues.
A CRO with a proven track record of proactively resolving issues and maintaining project timelines, even in the face of delays or unexpected variables, can be entrusted with confidence. It is also important to verify whether the CRO has provided close, hands-on support to clients with unique requirements, such as startups or small-to-mid-sized pharmaceutical companies.
Korea’s clinical trial infrastructure is world-class, and partnering with an experienced local CRO can accelerate the pace of development.
3. Data Management and Technical Capabilities
Data management capabilities and technical infrastructure are critical factors that determine the quality of modern clinical trials. When assessing a CRO, it is important to determine whether it actively utilizes modern tools such as electronic medical records (EMR), electronic data capture (EDC) systems, centralized databases, and remote monitoring technologies. Verify whether the CRO operates its own EDC system or employs validated commercial platforms, and whether proper validation procedures are in place to ensure data integrity.
Equally critical is the implementation of comprehensive data security and privacy measures, such as access controls, encryption, and regular backups. Given the volume and complexity of clinical trial data, selecting a CRO with strong technical proficiency, including expertise in statistical analysis software and pharmacovigilance (PV) systems—can significantly reduce errors and improve operational efficiency.
4. Effective Communication and Transparency
Effective communication and transparent operational practices are fundamental to establishing trust between a CRO and its sponsor. Evaluate whether the CRO consistently provides regular updates and promptly shares critical issues during the project. Confirm if a dedicated project manager (PM) serves as a centralized communication point and whether a clear escalation procedure is in place for urgent matters.
Reviewing past examples of how the CRO addressed challenges, such as delays in timelines or difficulties in patient recruitment can help gauge their responsiveness and reliability. A CRO that communicates openly and takes responsibility in managing issues demonstrates a collaborative culture, which increases the likelihood of successfully navigating unexpected circumstances.
5. Cost-Effectiveness and Transparent Quote
Cost is an essential consideration when selecting a CRO; however, choosing solely based on the lowest price can be risky. A comprehensive assessment of cost-effectiveness should begin with a review of the full scope of proposed services. Verify whether the CRO provides full-scope services—from initial clinical trial planning to regulatory submission preparation, monitoring, data management, statistical analysis, and final reporting and that any additional costs not included in the base estimate are transparently disclosed.
Even if the initial costs are somewhat higher, engaging an experienced CRO can shorten the overall development timeline and reduce rework, resulting in significant long-term cost savings. Conversely, selecting the lowest-priced proposal without sufficient experience may lead to operational inefficiencies or errors, ultimately causing higher expenses and time losses. Therefore, in addition to evaluating the reasonableness of the estimate, it is important to review the CRO’s performance track record (e.g., budget adherence, on-time delivery) to assess the value of the investment.
Careful consideration of all five factors: Experience, Expertise, Technical Capabilities, Communication, and Cost will facilitate the selection of a reliable CRO partner that most closely aligns with the specific requirements of the project.
Conclusion: Choosing the Right Partner for Clinical Trial Success
Choosing a CRO for your clinical trial is not just about cutting costs—it is a decision that can determine the success of your new drug development. By thoroughly evaluating potential partners against the key criteria outlined above, you can identify a CRO optimally suited to your project.
Collaboration with the right CRO can streamline trial management, accelerate development, and minimize risks. Korea is home to numerous CROs with capabilities on par with global standards. Notably, Intoinworld—with over 10 years of experience in both domestic and international trials—achieves high client satisfaction through its tailored services and transparent communication.
Looking for the ideal partner to ensure your clinical trial’s success? Intoinworld offers over a decade of specialized experience and a strong network, providing CRO services at global standards. If you are planning a clinical trial in Korea, contact us today for a consultation. With one-stop support from professional guidance to final reporting, we will partner with you throughout your new drug development journey. And if you don’t want to miss the latest pharma/biotech industry trends and clinical trial insights, consider subscribing to Intoinworld’s newsletter to get valuable information on a regular basis.
Frequently Asked Questions (FAQ)
Q1. Do I have to use a CRO for my clinical trial?
A: Legally, you are not required to use a CRO for a clinical trial, but most pharmaceutical and biotech companies choose to work with specialized CROs. Clinical trials involve complex procedures and strict regulations, so partnering with an experienced CRO can increase your success rate and shorten the development timeline.
If you try to manage a trial solely with in-house staff, the burden on your team and timeline is very high and mistakes are more likely. Therefore, working with a qualified CRO is generally a more efficient and effective approach.
Q2. Should I choose a global CRO or a domestic Korea CRO?
A: Global CROs have worldwide networks and standardized processes, giving them an edge in managing large multi-country trials, but they can be more costly. In contrast, domestic Korea CROs are familiar with local regulations and conditions and often offer greater cost efficiency. In Korea, roughly 70% of the market is served by large global CROs, while around 20–30 homegrown CROs handle the remaining 30%, each with their own strengths. You should select a partner based on your project’s scope and needs.
For example, if your trial will involve only Korean patients or is an early-stage single-country study, a local Korea CRO with deep local experience may provide more hands-on support. On the other hand, for a multinational trial or one requiring a global regulatory strategy, a global CRO is worth considering—just ensure they have a local office or a partnership in Korea to facilitate smooth execution.
Q3. What is the most important factor to consider when selecting a CRO?
A: It’s hard to single out just one factor, but we recommend prioritizing the CRO’s experience and expertise in your specific therapeutic area. The more trials a CRO has run in your field, the better prepared they will be to handle potential issues.
Additionally, you should evaluate their project management capabilities, data management technology, and communication style. In particular, check whether the CRO provides a transparent and detailed breakdown of their services and costs in their proposal—this can be a good indicator of their reliability and working approach. Ultimately, the key is to find a partner who can offer a tailored solution for your project and is easy to work with. If needed, consider inviting multiple candidate CROs for presentations or conducting audits to verify their capabilities before making your final decision.

