Introduction
Non Clinical Trials – the crucial preclinical stage of drug development – are undergoing a quiet revolution. Advanced AI technologies are transforming Non Clinical Trials by enabling researchers to predict drug behavior, identify toxicity, and optimize experiments in silico before ever entering a human study. This shift promises to accelerate timelines and cut costs by over 50% compared to traditional methods. In an industry where bringing a new therapy to market can take over a decade and cost upwards of $2 billion, such AI-driven efficiencies are a game-changer. Just as importantly, AI offers more ethical methods in Non Clinical Trials, reducing reliance on animal testing and aligning research with global ethical standards.
Understanding Non Clinical Trials and AI’s Role
Non Clinical Trials refer to all preclinical tests conducted before a drug or therapy is tested in humans. These include in vitro lab experiments, computer simulations, and in vivo animal studies to evaluate safety and biological activity. Traditionally, Non Clinical Trials are time-consuming and costly, as researchers must iterate through countless compounds and animal models to find a viable drug candidate.
Now, AI is turbocharging this process. By analyzing vast datasets – from genomic sequences to past experimental results – AI algorithms can predict which drug candidates are most likely to succeed long before traditional testing would reveal the answer. In essence, AI acts as a high-powered filter and guide, helping scientists focus on the most promising leads and design smarter Non Clinical Trials from the outset.
AI Applications Transforming Non Clinical Trials
AI is being applied across multiple fronts of Non Clinical Trials, fundamentally changing how early-stage research is done:
- Intelligent Drug Discovery: Machine learning models scan enormous chemical libraries to identify hits in silico. Instead of manually testing thousands of compounds, AI pinpoints which molecules have the features to interact with a disease target. This vastly speeds up the discovery phase of Non Clinical Trials. Notably, one AI platform moved a drug candidate to clinical testing in just 18 months versus the industry standard 42 months.
- Predictive Toxicology: An AI-driven approach can predict toxicity and side effects using algorithms trained on historical data. For example, companies use AI with “new approach methodologies (NAMs)” like organ-on-a-chip systems to simulate human organ responses. These models forecast how a drug might affect the body, flagging dangerous compounds early and reducing the need for animal subjects.
- Virtual Clinical Simulation: Before any human trials, AI can run virtual trial simulations. By modeling biological systems, AI helps design better Non Clinical Trials – selecting optimal doses and identifying biomarkers. It’s reported that AI-based simulations can cut trial duration by up to 30% through adaptive design, allowing researchers to condense what used to be sequential studies into parallel, faster investigations.
- Data Analytics & Decision Support: Non Clinical Trials generate huge amounts of data. AI’s ability to detect patterns means it can integrate multi-omics data (genomics, proteomics, etc.) to reveal insights that humans might miss. This guides scientists toward the most effective experiment next. AI also auto-analyses lab results, maintaining data integrity and highlighting significant findings in real time, thereby improving the quality of Non Clinical Trials outcomes.
Faster and More Cost-Efficient Development
By leveraging AI, pharma companies aim to compress the drug development timeline and budget like never before. A process that often spanned 10–15 years can potentially be shortened to 5–7 years. Likewise, overall R&D expenditures could drop significantly. Analysts project that AI could cut the time and cost of drug development by at least half. The impact is already evident in discovery and preclinical stages – for instance, AI predictions allow researchers to skip ahead to the most promising experiments, avoiding many dead-ends.
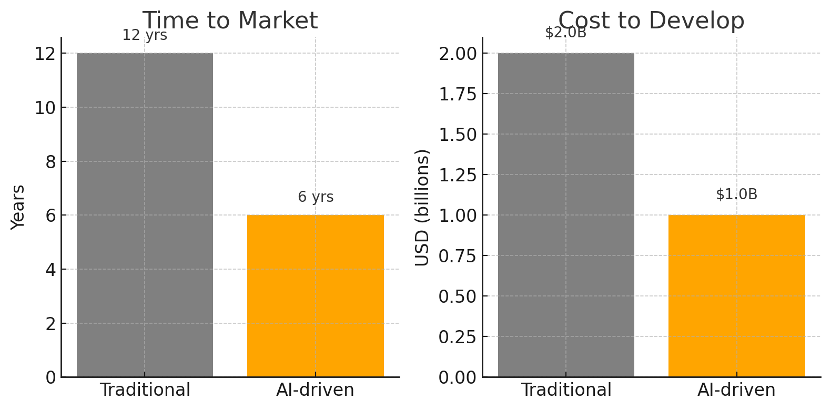
This drive for efficiency is not just theoretical. Globally, we see examples of how embracing technology improves development speed. In South Korea, for example, a strong digital infrastructure and innovative trial practices enable U.S.-level trial quality at roughly 60–70% of the cost. Such cost efficiency is achieved through streamlined operations and advanced data management. Non Clinical Trials stand to benefit from similar or greater cost savings as AI optimizes labor-intensive lab work and reduces costly late-stage failures. In practical terms, a drug candidate that might have required extensive animal testing and multiple revisions can reach First-in-Human trials much sooner and with leaner investment.
More Reading: Korea Clinical Trials Phase I/II Environment Procedures, Advantages, and Strategic Considerations
Ethical Advances and Reduced Animal Testing
One of the most profound benefits of applying AI to Non Clinical Trials is the ethical advantage. Traditional preclinical research often relies heavily on animal testing to gauge safety, which raises ethical concerns and practical limitations. AI offers a viable escape from this paradigm. Within the next few years, the FDA expects that animal studies will become the exception rather than the rule for safety testing, thanks in part to AI and other advanced models. By using AI-driven predictive models and human cell–based systems, researchers can replace or minimize animal use without compromising data quality. In fact, experts note that AI and new methods could cut preclinical timelines and costs by at least half while substantially reducing animal usage.
Beyond just reducing animal numbers, AI enhances the humane aspect of research. Complex algorithms and “digital twin” simulations can model human biology with increasing fidelity, meaning results are more directly relevant to humans and fewer sentient creatures are used. This shift aligns with the 3R principles (Replacement, Reduction, Refinement) in animal research and promotes public trust through more humane practices. As Patrick Smith of Certara observed, “we are getting to the point where we don’t actually need to do [animal testing] anymore”.
Such statements underscore a growing consensus that AI-enabled Non Clinical Trials can uphold rigorous ethical standards while still protecting patient safety and welfare. Importantly, oversight bodies like Institutional Review Boards (IRBs) and regulatory agencies are adapting to this new era, updating guidelines to incorporate AI-derived evidence as long as it meets validation and ethical review criteria.
Conclusion and Outlook
Non Clinical Trials are the bedrock of drug development – and with AI’s arrival, this foundation is becoming smarter, faster, and more humane. The integration of AI into preclinical research is no longer a futuristic concept but a present reality that biopharma professionals worldwide are embracing. AI algorithms now routinely assist in everything from candidate molecule selection to complex toxicity prediction. The results speak for themselves: shorter development cycles, reduced costs, and ethically responsible research. Importantly, this transformation is happening with careful oversight. Regulators are issuing guidelines for AI validation, and stakeholders are ensuring that human experts remain in the loop to interpret AI findings and maintain accountability.
For domestic and international biopharma teams, the message is clear – those who leverage AI in Non Clinical Trials will have a competitive edge. They can reach critical “go/no-go” decisions faster, allocate R&D budgets more efficiently, and enter clinical trials with greater confidence in their data. As we move forward, we can expect AI’s role to expand further, perhaps designing entire Non Clinical Trial programs autonomously or personalizing preclinical tests for specific patient subpopulations.
The future of Non Clinical Trials is a synergistic blend of human ingenuity and artificial intelligence, leading to safer, more effective treatments reaching patients sooner. In this new era of drug development, embracing AI is not just an option, but a strategic imperative.
Contact us for a free consultation and quote on integrating AI into your trials.
Interested in accelerating your preclinical research or need guidance on applying AI to Non Clinical Trials? Request a free quotation from our expert CRO team today. Our specialists will evaluate your project needs and propose a tailored plan to harness AI for better, faster Non Clinical Trial outcomes. We pride ourselves on delivering cost-effective, high-quality support, aligning with global standards and your specific goals. Don’t miss the opportunity to transform your R&D process – get your free quote now and let’s bring your innovation to the next level.
Stay informed on the latest trends shaping Non Clinical Trials and clinical development by subscribing to our newsletter. We provide regular updates on AI breakthroughs, regulatory changes, and best practices in biopharma R&D. Join a community of domestic and global professionals who get timely analysis and actionable insights delivered straight to their inbox. Subscribe now to ensure you never miss an update on how AI and other innovations are transforming the drug development landscape.
FAQs (Frequently Asked Questions)
Q1: What are Non Clinical Trials?
A1: Non Clinical Trials are the stage of drug development conducted before any tests on human volunteers. This phase includes laboratory experiments (in vitro), animal studies (in vivo), and computational simulations to assess a drug’s safety and efficacy profile. The goal of Non Clinical Trials is to gather enough evidence about a compound’s potential benefits and risks to justify moving forward to human Clinical Trials. In simpler terms, it’s everything a biopharma team does in R&D before testing a new drug in people.
Q2: How is AI improving Non Clinical Trials?
A2: AI improves Non Clinical Trials by making preclinical research faster, more accurate, and cost-efficient. Machine learning models can analyze large datasets to predict which drug candidates are worth pursuing, thus focusing resources on the most promising compounds. AI also simulates complex biological processes, helping scientists identify potential toxicity or efficacy issues early on. By reducing trial-and-error in the lab, AI shortens the Non Clinical Trials phase and increases the chances of success when a drug candidate moves to human testing.
Q3: Can AI reduce the need for animal testing in Non Clinical Trials?
A3: Yes. One of AI’s biggest contributions to Non Clinical Trials is the reduction of animal testing. AI-driven models (including computer simulations and cell-based assays) can predict how a drug will behave in humans, allowing researchers to skip or minimize certain animal experiments. Regulatory bodies are increasingly supportive of these methods – the FDA, for example, has a roadmap to make non-animal methods the default for toxicity testing within a few years. While animals are not completely phased out yet, AI has already significantly reduced the number of animals needed and is leading to more ethical, human-relevant research practices.
Q4: Are AI-assisted Non Clinical Trials accepted by regulators?
A4: Generally, yes – with proper validation. Regulatory agencies like the U.S. FDA and European EMA are actively developing guidelines for AI in drug development. They accept data from AI-assisted Non Clinical Trials as long as the methods are transparent, validated, and meet existing safety standards. For example, if an AI model is used to predict toxicity, regulators will want to see evidence that the model is accurate and reliable (often through a validation study). Agencies worldwide have even formed initiatives to fast-track promising methods (such as the FDA’s support for new approach methodologies). In short, regulators are cautiously optimistic: they welcome innovations that can improve efficiency or ethics in Non Clinical Trials, provided patient safety and scientific integrity are maintained.
Q5: What does the future hold for Non Clinical Trials with AI?
A5: The future of Non Clinical Trials with AI looks extremely promising. We expect to see more fully AI-driven laboratory automation, where robots and AI plan and execute experiments with minimal human input. Drug discovery might increasingly involve AI “colleagues” that design and test new molecules virtually. Also, personalized Non Clinical Trials could emerge – using AI, researchers might test drugs on virtual patient avatars or organ-on-chip systems tailored to specific genetic profiles, predicting which therapies work best for which populations before clinical trials begin.
Ultimately, AI will likely be standard in every preclinical toolkit, working hand-in-hand with scientists. This means safer drugs, developed faster, with less cost and fewer ethical compromises. The timeline to cure diseases could shrink dramatically, and patients will benefit from the accelerated arrival of new treatments.




