In 2025, the Korean MFDS approved 126 new Medical Device Clinical Trials, driven by a surge in digital health innovations and diverse therapeutic devices. This year-end analysis summarizes the Clinical Trials approvals, highlighting monthly trends, regulatory context, key sponsor activities, and technology implications for Medical Device Clinical Trials. The goal is to inform pharma/biotech professionals about Korea’s evolving Clinical Trials landscape and how Medical Device Clinical Trial approvals are shaping future innovation.
Summary Analysis of 2025 MFDS IND Approvals
From January 1 to December 31, 2025, the Ministry of Food and Drug Safety (MFDS) issued 126 approvals for Medical Device Clinical Trials (i.e., IND approvals specific to medical devices). This annual total of Clinical Trials approvals underscores Korea’s growing emphasis on medical device research. The monthly trend remained robust throughout the year, peaking in spring. Figure 1 below shows the monthly counts of MFDS-approved device Clinical Trials in 2025:
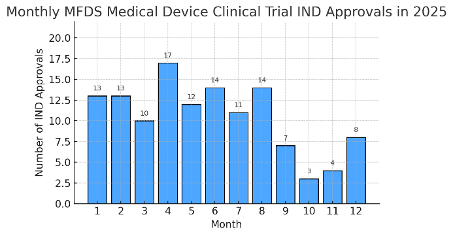
April saw the highest activity with 17 device Clinical Trials approved, coinciding with increased submissions after the Lunar New Year period. Early winter months (October–November) were relatively lower, possibly due to sponsors timing their Clinical Trials submissions before year-end. Overall, the monthly IND approval trend illustrates consistent MFDS review throughput, with no months dropping below 3 approvals. This stable cadence reflects Korea’s capacity to handle continuous Clinical Trials applications for medical devices.
Importantly, domestically developed devices comprised the majority of these Clinical Trials approvals. The introduction of Korea’s new Digital Medical Device Product Act in January 2025 significantly boosted the number of digital health device Clinical Trials. Local startups and medtech firms quickly leveraged the clearer regulatory pathway, resulting in a surge of digital therapeutics and AI-based device Clinical Trials. While multinational medtech companies were present, such as Medtronic’s trial of the Prevail drug-coated balloon aimed at reducing coronary restenosis, roughly three-quarters of the approved Medical Device Clinical Trials were sponsored by domestic organizations. This is a contrast to pharmaceutical IND trends where foreign-developed drugs dominate approvals. The rise of Korean digital health innovators has shifted the balance toward locally led Clinical Trials in the medical device sector.
The product types entering Clinical Trials in 2025 were diverse, ranging from rehabilitation software to high-end implants. Figure 2 illustrates the breakdown of approved trials by top product categories:
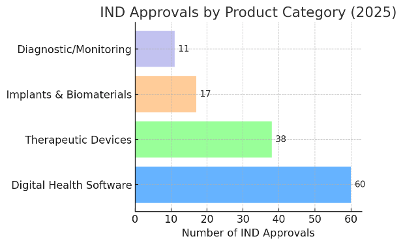
As shown above, digital health software devices accounted for the largest share of Clinical Trials approvals. Approximately 45–50% of all device Clinical Trials were for Software as Medical Device (SaMD) products – including digital therapeutics and AI-driven diagnostic aids. Traditional therapeutic devices (lasers, stimulators, physical therapy equipment, etc.) formed the second-largest group, at roughly one-third of approvals. Implantable devices and biomaterials (such as orthopedic implants, cardiac valves, and tissue regeneration materials) made up about 10–15%, while diagnostic and patient-monitoring devices comprised the remainder (under 10%). This distribution highlights how software-based Medical Device Clinical Trials have nearly overtaken hardware trials in volume, marking a pivotal shift in the Clinical Trials landscape for devices.
Regulatory Context and IND Approval Process
Korea’s regulatory framework for Medical Device Clinical Trials is rigorous and evolving. The MFDS reviews each device trial under an Investigational Device Exemption (IDE) system to ensure trial safety, scientific validity, and ethics. No Clinical Trial can proceed without MFDS approval of the plan and a parallel Institutional Review Board (IRB) clearance. In 2025, the regulatory context was notably shaped by the Digital Medical Products Act (DMPA), implemented in January. This new law established explicit requirements for digital health device Clinical Trials – from submission procedures and quality control to post-approval surveillance. By formalizing standards for software and AI-based medical devices, the MFDS provided clarity that encouraged more sponsors to initiate Clinical Trials in emerging areas like digital therapeutics.
Under MFDS oversight, sponsors must submit detailed investigational plans, including preclinical data, risk analyses, and clinical protocols. The review timelines for device Clinical Trials are typically efficient – often around 30 working days for straightforward cases – reflecting MFDS’s commitment to fostering innovation while upholding safety. In 2025, MFDS also issued new guidelines, such as those for VR/AR-based digital devices, to help sponsors align their Clinical Trials with regulatory expectations. These guidelines address issues like algorithm validation, cybersecurity, and data privacy, ensuring that advanced technology Clinical Trials meet global Good Clinical Practice (GCP) standards.
Another regulatory development was the emphasis on post-market considerations even at the trial stage. For instance, sponsors of digital health Clinical Trials are now expected to have plans for ongoing software updates and real-world performance monitoring after approval. MFDS’s proactive stance means that companies designing Clinical Trials for medical devices – especially connected or AI-powered devices – must incorporate lifecycle management and evidence generation strategies early on. Overall, Korea’s regulatory environment in 2025 combined speed with stringency, facilitating a high volume of Medical Device Clinical Trials without compromising patient protections.
Sponsor Activity and Key Approvals
The surge in device Clinical Trials was led by a mix of innovative startups, research institutions, and a few global players. Top sponsor companies distinguished themselves by the number of IND approvals obtained for their devices. According to MFDS data, the most active sponsor received 10 Clinical Trial approvals in 2025 for a single product – a rehabilitation medical software – reflecting an extensive development program. Another sponsor secured 9 Clinical Trial approvals for a cognitive therapy software targeting neurological conditions. Table 1 summarizes the leading sponsors (by proxy of their product approvals):
| Sponsor (Product) | IND Approvals |
| Domestic Digital Therapeutics Firm – rehab software | 10 Clinical Trials |
| Domestic Digital Therapeutics Firm – cognitive software | 9 Clinical Trials |
| Medtech Company – physical therapy device | 6 Clinical Trials |
| Digital Health Startup – mental health software | 4 Clinical Trials |
| Biomaterials Company – tissue regenerative filler | 4 Clinical Trials |
Table 1: Top sponsors by number of MFDS-approved Medical Device Clinical Trials in 2025. (Sponsor names are represented by product type for confidentiality.)
These figures show that Korean digital health companies were especially prolific – a single digital therapeutics developer accounted for nearly 8% of all device Clinical Trials approved. Rehabilitation-focused software (for example, programs aiding recovery of mobility or function) saw multiple trials by one sponsor, indicating a broad effort to validate its efficacy across conditions and settings. Similarly, a cognitive therapy software (addressing cognitive impairment or dementia) was the subject of 9 trials by its sponsor, likely including pilot studies and confirmatory trials to pave the way for marketing approval.
Traditional device manufacturers were less dominant in count but still notable. A domestic company developing an innovative physical therapy instrument (possibly a robotic rehabilitation device) obtained 6 trial approvals, highlighting the continued importance of hardware solutions in rehabilitation Clinical Trials. Sponsors focusing on mental health digital interventions (e.g. a digital therapy for depression or addiction) and tissue-engineered biomaterials (such as dermal fillers or regenerative scaffolds) each received 4 approvals. It’s worth noting that some global medtech sponsors were active as well – for instance, Medtronic’s Korea arm conducted trials for its Prevail™ drug-coated balloon and a peripheral vascular stent graft, leveraging Korea’s Clinical Trials infrastructure for global studies. However, no single multinational outpaced the top domestic players in sheer number of device Clinical Trials in 2025.
Beyond the raw counts, the breadth of indications covered by these sponsors is striking. The approved device trials targeted oncology, cardiology, neurology, metabolic diseases, and more. For example, one sponsor’s approvals included an AI-driven imaging software for stroke diagnosis (highlighting a focus on acute neurology), while another’s included a closed-loop insulin delivery software (an “artificial pancreas”) for type 2 diabetes. This spread underscores that Korea’s medtech sponsors are exploring diverse fields through Clinical Trials, from hospital-based high-tech interventions to at-home digital therapies.
Technology Trend Implications
The 2025 MFDS approvals for Medical Device Clinical Trials reveal critical technology trends shaping the industry:
- Explosion of Digital Health and AI: Over 40% of approved Clinical Trials were for digital health technologies, confirming that software-based medical devices have become mainstream in Korea’s clinical research. These include digital therapeutics (DTx) aiming to treat conditions like insomnia, depression, substance use disorder, and mild cognitive impairment via software interventions. The MFDS approvals spanned apps and software using cognitive behavioral therapy principles, VR-based treatments for chronic insomnia, and motivational digital programs for addiction recovery. The implication is that Korea is embracing non-pharmacological treatments delivered through software – a trend likely to complement or compete with traditional drug therapies. Additionally, AI-powered diagnostic software won several approvals, such as algorithms analyzing medical images (CT, MRI, endoscopy) to assist in detecting strokes or cancers. These Clinical Trials aim to validate AI tools like JLK-CTI for stroke CT analysis and “SHERPA RCC” for kidney tumor risk on CT, potentially accelerating the integration of AI into routine clinical practice. Korea’s strong IT infrastructure and the new DMPA law are facilitating rapid evaluation of such digital innovations in controlled Clinical Trials.
- Personalized and Precision Devices: Another trend is the move toward personalized medical devices, exemplified by Clinical Trials of 3D-printed implants and patient-tailored therapy. Notably, MFDS approved a trial for a custom 3D-printed artificial ankle joint implant, designed to match an individual patient’s talus bone for revision surgery. This reflects the rise of precision orthopedics – leveraging 3D printing to improve fit and outcomes. The trial’s significance lies in demonstrating improved alignment and reduced complications compared to standard implants, a step toward mainstreaming personalized implants. Likewise, patient-specific biomaterials (e.g. customized tissue fillers) were tested in head-to-head trials against market leaders. The ability to compare a novel filler with an established product like Juvederm® in a controlled Clinical Trial indicates a maturing biomaterials sector aiming to prove equivalence or superiority. The trend suggests Korea is nurturing advanced manufacturing (3D printing, novel biomaterials) combined with Clinical Trials to validate personalized treatments.
- Connected and Closed-Loop Systems: Several 2025 approvals highlight the convergence of devices with digital connectivity for real-time monitoring or automated therapy. The trial of “CloudLoop” insulin delivery software is a prime example – it integrates a continuous glucose monitor with an insulin pump in a closed-loop, using algorithms to adjust insulin dosing automatically (an AI-driven artificial pancreas). The MFDS’s approval of this exploratory trial signals regulatory support for complex IoT (Internet of Things) medical systems and foreshadows more “smart” Medical Device Clinical Trials (e.g. closed-loop ventilators or neurostimulators with feedback control). Additionally, wearable monitors (like a wearable skin thermometer and vital sign oximeters) gained approvals, showing interest in remote patient monitoring solutions. These devices align with global telehealth trends and Korea’s push for digital healthcare integration. The implication is that future Clinical Trials will increasingly evaluate not just standalone devices, but systems of devices + software + AI that together deliver personalized therapy.
- Non-invasive and Novel Therapies: The range of MFDS-approved trials also underscores a tilt toward less invasive treatment modalities. Energy-based therapies were prominent, including focused ultrasound (both high-intensity for ablation and low-intensity for neurostimulation) and low-level laser therapy devices. For instance, a trial of a focused ultrasound stimulation system for neurological disorders was approved, as was a trial of a transcranial direct current stimulation (tDCS) device for psychiatric therapy. These modalities represent cutting-edge, non-invasive treatments for conditions like depression or epilepsy (e.g. a vagus nerve stimulator for epilepsy was among approvals). Their presence in multiple Clinical Trials indicates a trend toward exploring alternatives to surgery or drugs by using device-delivered energy to modulate physiology. If successful, such trials could broaden therapeutic options in neurology and rehabilitation, reinforcing Korea’s role in advancing novel device therapies.
In summary, the 2025 Clinical Trials approval cohort paints a picture of a dynamic medical device R&D ecosystem. Korea is validating digital treatments for chronic diseases, testing AI as a co-pilot in diagnostics, pioneering patient-customized implants, and investigating next-gen therapeutic devices – all under the watch of a supportive regulatory framework. These trends not only bolster Korea’s status as a global hub for Clinical Trials but also promise to impact healthcare delivery by introducing new modalities that are tested and proven locally. The broad scope of trials (from hospital ICU decision-support software to consumer-facing mental health apps) implies that Medical Device Clinical Trials are increasingly multidisciplinary, often requiring collaboration between software engineers, AI scientists, clinicians, and manufacturers.
Conclusion and Call to Action
Korea’s 2025 MFDS approvals for Medical Device Clinical Trials signal a new era of innovation, with digital health at the forefront and a strong pipeline of both local and international projects. The year’s 126 approvals represent not just a quantity, but a qualitative shift toward smart, patient-centric technology in Clinical Trials. For domestic and global pharma/biotech professionals, these trends highlight opportunities to leverage Korea’s conducive environment for cutting-edge device research – from AI diagnostics to DTx and personalized implants. MFDS’s efficient yet thorough regulatory process, combined with initiatives like the DMPA, ensure that approved Clinical Trials meet high standards, thereby de-risking subsequent market approvals.
For companies planning their own Medical Device Clinical Trials, staying abreast of Korea’s regulatory guidelines and approval trends is crucial. As seen in 2025, aligning trial design with MFDS priorities (e.g. data quality for AI, post-market surveillance plans, comparative effectiveness for novel materials) can accelerate approval. Engaging with local CROs or consultants experienced in Korea’s device trials can further streamline the process.
Need support with your clinical trial strategy? We offer expert services to help navigate MFDS approvals and design efficient Medical Device Clinical Trials. Please request a quote for personalized consulting on regulatory submissions, trial management, or market entry in Korea. To stay informed on the latest MFDS Clinical Trials news and insights, subscribe to our newsletter – delivered monthly with analysis of IND approvals and policy updates. Visit our homepage to learn more about our offerings and success stories. We are ready to partner with you to advance your medical device development through successful Clinical Trials in Korea.



