Overview
In 2025, the Korean Ministry of Food and Drug Safety (MFDS) granted 636 Investigational New Drug (IND) approvals, reflecting sustained high Clinical Trials activity. Monthly approvals fluctuated from a peak of 82 IND approvals in January to a low of 31 in November, indicating some seasonal and operational variability. Overall, the volume remained robust throughout the year, underscoring Korea’s reliability as a prime location for Clinical Trials. Oncology emerged as the dominant therapeutic area, and roughly 70% of all IND clearances were for treatments developed by international sponsors – reaffirming Korea’s role as a global hub for Clinical Trials. The original MFDS IND approval data for 2025 is available as a downloadable Excel file on our website, allowing interested professionals to examine the detailed breakdown.
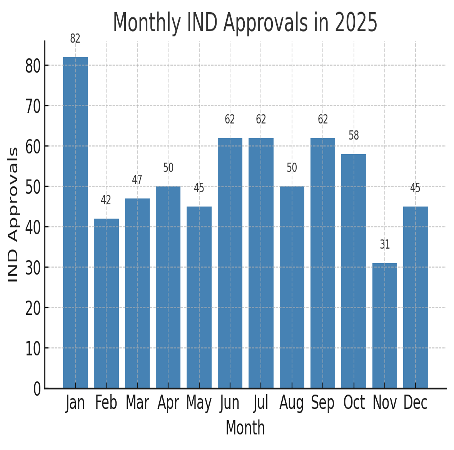
Monthly MFDS IND approvals in 2025. The bar chart illustrates MFDS-approved INDs per month, totaling 636 for the year. Approvals spiked in January (82), possibly due to sponsors accelerating submissions after year-end, and remained in the 40–62 range most months. A slight dip occurred in early Q2 (45 in May) followed by a mid-year surge (62 in June, 62 in July). Notably, November was the slowest month (31), before activity rebounded in December (45). This trend suggests that while Korea’s Clinical Trials pipeline is consistently active, it can be influenced by external timing factors such as regulatory cycles or sponsor strategy. Importantly, even the lowest month (November) saw over 30 new Clinical Trials start – a figure many countries would envy – highlighting the country’s strong baseline of research activity.
IND Approvals by Phase
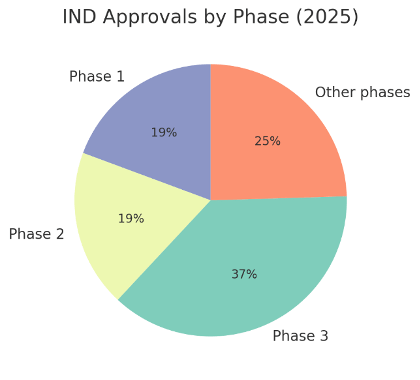
IND approvals by phase in 2025. The majority of 2025 IND approvals were for late-phase trials, emphasizing Korea’s attractiveness for pivotal studies. Phase 3 trials accounted for the largest share (roughly 37% of approvals), reflecting many programs reaching efficacy confirmation stages. A significant portion were Phase 1 trials (~19%) and Phase 2 trials (~19%), indicating a healthy influx of early-stage studies to fuel the pipeline. The remaining ~25% were in adaptive or combination phases (e.g. Phase 1/2, Phase 2/3 bridging studies, Phase 3b extensions, as well as a few Phase 4 post-market trials). This distribution – a high number of Phase III INDs alongside numerous Phase I and II – demonstrates that Korea supports the full spectrum of Clinical Trials, from first-in-human safety studies to large-scale international trials. The strong showing of Phase III suggests many drug developers (especially global ones) trust Korean sites for crucial late-stage trials. At the same time, the volume of Phase I/II approvals and other hybrid designs points to innovative trial strategies (such as seamless trials and dose-escalation studies) being pursued in Korea’s regulatory environment.
Early-phase trials (Phase 1 and 2) together made up roughly 42% of approvals, which aligns with global sponsors increasingly including Korea in Phase I studies. In fact, recent analyses noted a notable increase in multinational Phase I Clinical Trials in Korea, contributing to the country’s growing share of early development work. Meanwhile, late-phase (Phase 3) dominance in the IND mix underscores that many therapeutics tested in Korea are on the cusp of regulatory submission. The presence of Phase 3b and Phase 4 trials – though only a handful of cases – shows that sponsors also use Korea for extension studies and post-approval research, further cementing the nation’s end-to-end Clinical Trials appeal.
Domestic vs. Global Development
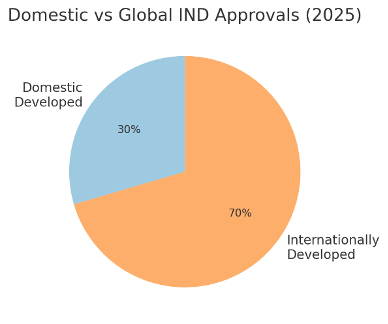
Ratio of domestic vs. internationally developed IND approvals in 2025. Out of 636 IND approvals, roughly 70% were for drug candidates developed overseas, while about 30% were for compounds from domestic Korean developers. This 70:30 split in favor of international programs is consistent with recent years and highlights South Korea’s continued attractiveness to multinational pharmaceutical companies. Global firms frequently choose Korea as a site for their trials due to the country’s efficient regulatory process and high-quality medical infrastructure. By contrast, local Korean biopharma companies contributed nearly a third of INDs – a substantial share that reflects a vibrant home-grown pipeline, albeit weighted more toward early-stage trials.
One reason for the dominance of foreign-developed drug trials is regulatory and logistical: overseas sponsors often partner with domestic contract research organizations (CROs) to navigate the MFDS process. Engaging a Korean CRO and principal investigators allows multinationals to leverage Korea’s fast review timelines (MFDS reviews IND applications in ~30 working days) and access diverse patient populations. These collaborations have made Korea a strategic location for late-phase Clinical Trials – many of the foreign INDs in 2025 were global Phase III studies seeking to include Korean patients and sites.
Meanwhile, domestic IND approvals in 2025 were largely concentrated in Phase 1 trials initiated by biotech startups and research institutes. Korean companies like Celltrion and Hanmi are using INDs to push novel therapies (e.g. antibody-drug conjugates, biosimilars, and cell therapies) into first-in-human studies. This pattern mirrors 2024, where local firms predominantly obtained IND clearance for early trials while foreign sponsors accounted for most Phase 3 trials in Korea. The disparity suggests that while Korea’s indigenous R&D is growing, many homegrown drugs are still in proof-of-concept stages, whereas later-phase Clinical Trials in Korea often involve drugs originally developed abroad.
Encouragingly, the 30% domestic share still translates to nearly 200 locally developed INDs – a volume on par with or higher than many larger markets’ total trial count. It signifies an expanding contribution of Korean companies to the clinical development landscape. Government initiatives such as increased R&D funding and fast-track designations for domestic innovative drugs are expected to further boost local IND numbers in coming years, gradually balancing the foreign-domestic ratio.

Leading Sponsors and Company Contributions
The 2025 IND approval data reveals interesting dynamics among sponsoring organizations. Global CROs and pharma affiliates were prominent repeat IND filers: for example, IQVIA Korea and Parexel Korea each handled around 25–26 IND approvals, the highest of any entities. They were closely followed by top multinational drug companies’ local units like MSD Korea (20 INDs), AstraZeneca Korea (~15), Novartis Korea (~14), and Sanofi-Aventis Korea (~9). These figures underscore how actively global firms are using Korea for their Clinical Trials – whether directly via their subsidiaries or indirectly through CRO partners. The leading global sponsors focused on a mix of late-stage trials (especially oncology and immunology) and some mid-phase studies, leveraging Korea’s patient pools for complex trials such as immuno-oncology combinations and rare disease therapies.
In contrast, domestic companies accounted for fewer INDs individually, but a couple of local players stood out. Celltrion, one of Korea’s biopharma leaders, secured around 10–11 IND approvals in 2025, the most among domestic sponsors. These included trials for innovative biologics like tissue-factor targeting ADCs and biosimilars, reflecting Celltrion’s aggressive pipeline advancement. Daewon Pharmaceutical also obtained multiple INDs (around 7) for new gastrointestinal treatments (such as novel acid blockers), one of which is a large Phase 3 trial involving hundreds of patients in gastroesophageal reflux disease. Other notable domestic IND sponsors were SK Plasma (for a Phase 3 immunoglobulin trial in immunodeficiency), Chong Kun Dang, EHL Bio, and Oscotec, all of whom launched Phase 1 or 2 studies for novel drug candidates.
It’s worth noting that several investigator-initiated trials by top hospitals and research centers (e.g. Asan Medical Center, National Cancer Center) also contributed to the IND approvals count. While not company-sponsored, these institution-led studies – often classified under “domestic” – targeted areas like surgery adjuncts and repurposing existing therapies, adding to the diversity of Korea’s Clinical Trials portfolio. For instance, the National Cancer Center ran a Phase 2 trial on a fluorescent imaging agent for gastric cancer surgery. This investigator-driven research complements industry trials and showcases Korea’s academic sector strength.
Overall, the mix of sponsors in 2025’s IND approvals highlights a collaborative ecosystem: global pharma and CROs bring large-scale trials and resources, while domestic firms and investigators introduce innovation and local expertise. This synergy further solidifies Korea’s reputation in the international Clinical Trials community.
Therapeutic Area Trends
As in prior years, oncology was the leading therapeutic area for IND approvals in 2025 – a trend consistent with Korea’s prominence in cancer research trials. By our analysis, oncology trials accounted for nearly half of all INDs (spanning solid tumors, hematologic malignancies, and immuno-oncology agents). A review of trial titles confirms that a large number of approvals were for targeted cancer therapies, immunotherapies (including several CAR-T cell trials) and novel combination regimens in oncology. This aligns with global R&D priorities and demonstrates that Korea remains deeply involved in cutting-edge cancer Clinical Trials. Notably, sponsors pursued not only late-stage oncology trials in Korea but also early-phase oncology studies to evaluate first-in-class agents in Korean patients.
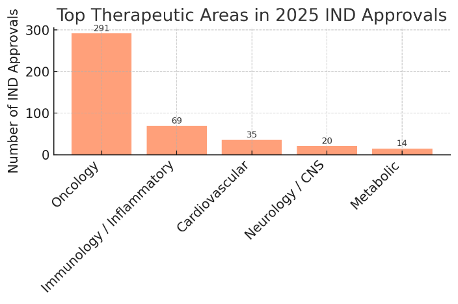
Top therapeutic areas by number of IND approvals in 2025. After oncology, the next most common therapeutic areas were immunology/inflammatory diseases and rare diseases. Approximately 10–12% of INDs targeted immunological conditions – for example, autoimmune disorders, inflammatory bowel diseases (Crohn’s, ulcerative colitis), and various forms of arthritis and dermatitis benefited from new clinical studies. Several INDs for atopic dermatitis, lupus, and rheumatoid arthritis reflect the interest in immune-modulating therapies. There was also a cluster of trials in rare diseases, ranging from genetic metabolic disorders to rare pediatric conditions, underscoring a commitment to orphan drug development (even if each specific rare condition had only one trial, collectively they form a notable segment).
Cardiovascular and metabolic disorders formed another significant category, though smaller in share. Roughly 5–6% of 2025 IND approvals were in cardiovascular diseases (including heart failure, hypertension, and stroke prevention trials), and about 2–3% were in metabolic/endocrine conditions (such as diabetes and obesity). For instance, an IND for maridebart cafraglutide, a novel dual agonist for obesity and atherosclerosis, and another for an innovative insulin-adjunct in diabetes were among the high-profile metabolic trials. In cardiology, an IND for BAY 94-8862 (finerenone) targeting heart failure was approved, reflecting the continued interest in cardiorenal outcomes trials.
Outside of these main areas, the remaining ~25% of trials spanned a broad spectrum of fields. Korea’s 2025 INDs included studies in neurology/CNS (e.g. Alzheimer’s disease, Parkinson’s, schizophrenia – about 3% of approvals), infectious diseases (including a few COVID-19 vaccine boosters and antiviral trials, plus studies in HIV and hepatitis – about 1–2% of approvals), and others like ophthalmology, gastroenterology (non-inflammatory GI disorders), nephrology, and dermatology. Noteworthy examples are a long-term trial of a novel therapy for idiopathic pulmonary fibrosis, and a Phase 3 vaccine trial for an emergent viral illness, reflecting responsiveness to public health needs. The diversity of these “other” trials indicates that Korea’s Clinical Trials portfolio is well-rounded and not limited to any single domain, even though oncology clearly dominates.
Crucially, many of the innovative modalities (gene therapies, cell therapies, etc.) are embedded within these therapeutic areas. For example, 2025 saw multiple cell therapy INDs – CAR-T cell trials for cancer, an adipose-derived stem cell therapy IND for atopic dermatitis, and an umbilical cord MSC-derived exosome therapy IND for ischemic stroke – highlighting that Korea is embracing advanced biotherapeutics across different diseases. Such trials point to the future of Clinical Trials in Korea, where advanced therapies and precision medicine play an increasingly prominent role.
Korea’s Position in Global Clinical Trials
The 2025 IND approval trends reinforce South Korea’s position as a leading Asian hub for Clinical Trials and a key player globally. The high volume of foreign-sponsored trials (448 INDs from global developers) demonstrates strong international confidence in Korea’s regulatory system and clinical infrastructure. Korea offers sponsors rapid review timelines, alignment with ICH GCP standards, and access to experienced investigators and large, well-organized hospitals. These advantages have placed Korea consistently among the top countries for industry-sponsored Clinical Trials – it ranked 4th worldwide in number of clinical trials in 2023, with Seoul being one of the busiest cities for trials.
At the same time, the significant participation of domestic companies (nearly 30% of INDs) and investigators shows Korea’s internal biopharma ecosystem is flourishing. Local biotechs are increasingly launching innovative trials, and government support (via the Korea National Enterprise for Clinical Trials, KoNECT, and regulatory incentives) is fostering more home-grown innovation. The MFDS has been implementing regulatory improvements – such as shorter IND review times and specialized guidelines for advanced therapies – to maintain a competitive edge and encourage both foreign and domestic research. These efforts help ensure that Korea remains an attractive location for Clinical Trials across all development stages.
In summary, the MFDS IND approvals of 2025 highlight a vibrant and internationally connected clinical research environment in Korea. Oncology trials led the charge, but a wide array of therapeutic areas were represented, reflecting the country’s diversified research focus. The dominance of global-sponsor trials underscores Korea’s integration into multinational development strategies, while the steady flow of domestic trials signals rising local innovation. For pharmaceutical and biotech professionals, these trends affirm that conducting Clinical Trials in Korea can offer both speed and quality – whether it’s a large Phase III program for a new oncology drug or an early-phase feasibility study for a novel biotech molecule. With its blend of efficient regulation, skilled clinicians, and commitment to innovation, South Korea is poised to continue its growth as a pivotal hub for Clinical Trials in the Asia-Pacific region and beyond.


