Overview
In December 2025, the Ministry of Food and Drug Safety (MFDS) approved eight Medical Device Clinical Trial applications, marking a busy end to the year. These MFDS approvals for Medical Device Clinical Trial proposals spanned a diverse range of technologies and therapeutic areas. Notably, a significant number of the Medical Device Clinical Trial projects focused on neuromodulation (such as focused ultrasound and vagus nerve stimulation) and digital health innovations (including AI-driven diagnostics and digital sensory devices).
This breadth underscores Korea’s growing emphasis on cutting-edge medical device development and the robust support for Medical Device Clinical Trial activity in the country. The MFDS’s efficient regulatory process – typically completing review of a Medical Device Clinical Trial application within ~30 working days– continues to attract both domestic and international sponsors to conduct trials in Korea.
To illustrate December’s landscape, the approved Medical Device Clinical Trial projects included: a focused ultrasound system (“NMS-01”) aiming to temporarily open the blood-brain barrier for chemotherapy delivery in brain tumor patients, a digital olfactory test device (“DS-ODTD-01”) for evaluating smell function, an AI-powered cardiac MRI software (“Myomics DX”) to assist diagnosis of cardiac amyloidosis, a transcranial magnetic stimulation device for cognitive impairment therapy, a photoacoustic vascular imaging device (“Perigio”) for peripheral artery disease, a high-intensity focused ultrasound device (“LIPOGE-X”) for aesthetic skin tightening, a vagus nerve stimulation device (“NQ-VNS”) for chronic dizziness, and a non-invasive ear neuromodulation device (“Soricle”) for tinnitus relief.
Each of these Medical Device Clinical Trial approvals represents a strategic advance in its field – from addressing unmet clinical needs (e.g. glioblastoma therapy, tinnitus treatment) to expanding the scope of digital therapeutics and diagnostics.
IND Approvals by Product Category
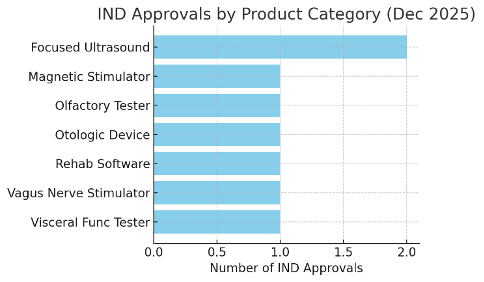
The December Medical Device Clinical Trial approvals can be categorized into seven product types. As shown in Figure 1, Focused Ultrasound systems led the month’s count with 2 approvals (targeting oncology and cosmetic indications respectively). This suggests concentrated interest in ultrasound-based Medical Device Clinical Trial innovations, both for treating serious diseases and for aesthetic medicine. The other categories – including Olfactory test devices, Rehabilitation software, Magnetic stimulators (transcranial magnetic stimulation devices), Visceral function testers (e.g. a photoacoustic imaging device for vascular diagnostics), Vagus nerve stimulators, and Otologic devices for ear disorders – each accounted for one approved Medical Device Clinical Trial project.
The even distribution among these diverse categories highlights the breadth of Korea’s medical device R&D: from digital diagnostic tools to therapeutic energy devices. Notably, software and AI-based medical devices continued to feature prominently, reflecting a global trend of software-driven innovation in Medical Device Clinical Trial approvals. The presence of two Medical Device Clinical Trial approvals in the focused ultrasound category (covering both a brain therapy device and a skin therapy device) also underscores how versatile platform technologies are being explored across multiple medical applications.
Top Companies by Approvals
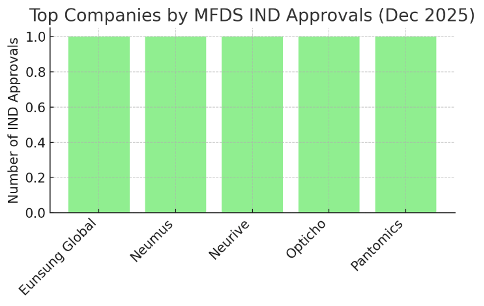
During December 2025, five different companies each secured MFDS approval for one Medical Device Clinical Trial. Figure 2 lists the leading industry sponsors: Eunsung Global, Neumus, Neurive, Opticho, and Pantomics all had one Medical Device Clinical Trial approval each.
This indicates a highly diversified sponsor landscape – no single company dominated the month’s Medical Device Clinical Trial approvals. Notably, these companies represent various niches in the medtech sector: for example, Eunsung Global’s approval was for LIPOGE-X, a novel HIFU aesthetic device, while Neumus’s approval was for NMS-01, an ultrasound-based system aimed at brain cancer therapy (an MFDS-designated innovative device). Neurive obtained an approval for Soricle, a non-invasive vagus nerve stimulator targeting tinnitus (Soricle is a Class III electroceutical device developed in Korea).
Opticho’s approval concerned Perigio, a photoacoustic imaging device that visualizes peripheral blood vessels without contrast agents. Pantomics received approval for the Myomics DX AI software, underlining the rise of AI in Medical Device Clinical Trial design. In addition to industry sponsors, it should be noted that three of the eight December approvals were investigator-initiated Medical Device Clinical Trial projects led by hospital researchers (for instance, the digital olfaction test and the vestibular VNS trial were driven by academic medical centers).
The strong showing of investigator-led trials alongside company-sponsored trials emphasizes the collaborative ecosystem in Korea’s clinical research arena – academia and industry are both actively contributing to Medical Device Clinical Trial activity.
Class IV Device Trial Trend
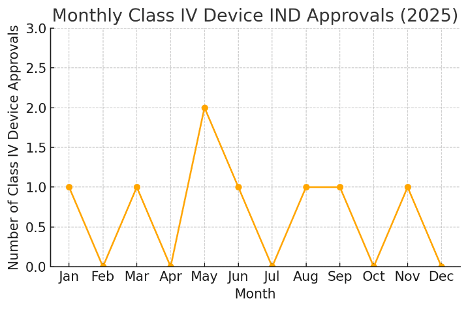
Interestingly, December 2025 had no Class IV Medical Device Clinical Trial approvals (Class IV being the highest-risk medical devices requiring the most stringent oversight). Figure 3 illustrates the monthly trend of Class IV device trial approvals throughout 2025. While earlier months in 2025 saw a handful of Class IV device trials (e.g. implantable or life-sustaining devices) approved by MFDS, the absence of Class IV approvals in December is notable. This could suggest that December’s surge in Medical Device Clinical Trial activity was driven largely by low-to-moderate risk innovations (Class II and III devices such as software, diagnostic equipment, and external stimulators), rather than by high-risk implantable devices.
The Medical Device Clinical Trial focus at year-end appears to lean toward technologies that are innovative yet relatively less invasive. It’s possible that some Class IV device submissions were finalized earlier in the year, or will resume in the new year, resulting in this December dip. Overall, the fluctuating monthly pattern of Class IV trial approvals underscores the adaptive regulatory pipeline – heavier one month, lighter the next – depending on when complex devices reach the IND stage. For regulatory professionals, this trend highlights the importance of timing in submission strategies for high-risk medical devices.
Conclusion and Industry Outlook
December 2025’s Medical Device Clinical Trial approvals showcase South Korea’s dynamic regulatory environment and commitment to fostering innovation in medical devices. The mix of approvals – from advanced therapeutic devices (ultrasound and electromagnetic stimulators) to AI-driven diagnostics and digital health tools – indicates that Korea continues to embrace a wide spectrum of new technologies through the MFDS clinical trial approval process.
Each approved Medical Device Clinical Trial in this month contributes to a broader narrative of medical innovation: for example, ultrasound-mediated blood-brain barrier opening could revolutionize neuro-oncology treatment, and non-invasive neuromodulation devices offer new hope for chronic conditions like tinnitus and balance disorders. The active participation of both medtech companies and clinical investigators in these trials reflects a collaborative ecosystem aimed at accelerating medical advancements.
Korea is increasingly recognized as a global hub for clinical research, supported by MFDS’s systematic and swift review procedures. Medical device developers can leverage this favorable environment to conduct efficient Medical Device Clinical Trial projects, gather high-quality data, and expedite their path to market. The strategic insights from December 2025 underscore that staying informed of regulatory trends is crucial for success in this landscape. Our platform offers expert regulatory consulting and the opportunity to request a quote for guidance on MFDS submissions.
We also provide up-to-date industry news and regulatory changes – you can subscribe to our newsletter to receive these Medical Device Clinical Trial updates. For comprehensive support in your clinical development journey, feel free to visit our homepage. By capitalizing on timely information and professional guidance, CROs and device developers can navigate the Korean Medical Device Clinical Trial landscape effectively and turn regulatory compliance into a strategic advantage.



