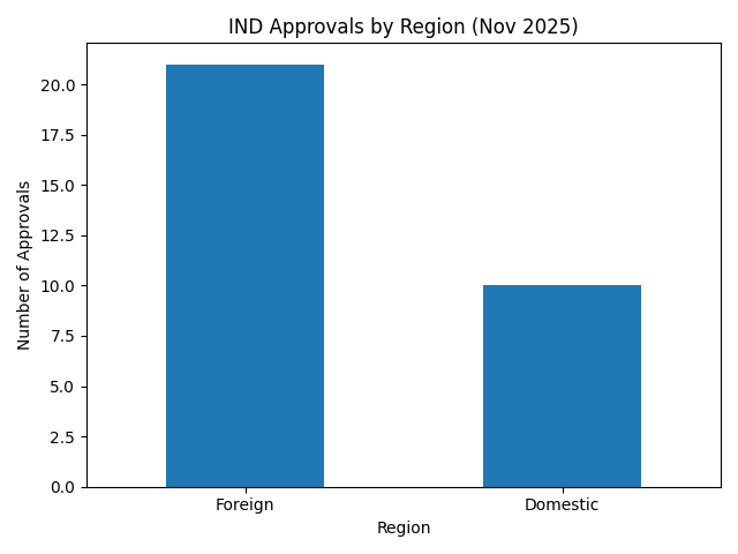
A review of the MFDS IND approval status for November 2025 shows that 31 clinical trial approvals were granted. Although this is a significant drop from 58 approvals in the previous month, it demonstrates that Korea continues to play a vital role as a global hub for clinical trials. Of the approvals, 10 (about 32 %) were for drugs developed domestically and 21 (about 68 %) were for drugs developed overseas. This mix of domestic and foreign trials reflects international confidence in Korea’s regulatory environment and clinical infrastructure.
Approvals by Region
Looking at approvals by region, 21 of the drugs were developed abroad, far outnumbering the 10 domestically developed drugs. Foreign companies are leveraging Korea’s rapid review procedures and robust research environment to conduct late‑stage clinical trials, while domestic companies are concentrating on early‑stage studies to confirm safety and dosing. The bar chart below compares the number of domestic and foreign IND approvals.
Approvals by Phase
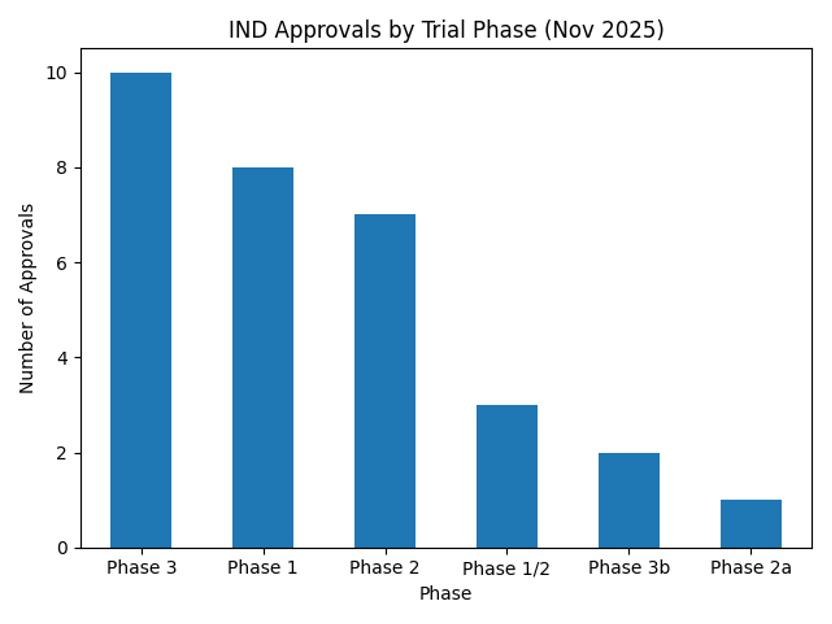
By phase, Phase 3 trials accounted for 10 approvals, while Phases 1 and 2 each had eight and seven approvals, respectively. Foreign drugs are heavily weighted toward Phase 3 and Phase 2 approvals, indicating a focus on later‑stage trials, whereas the majority of domestic drugs were in Phase 1. The following chart visualizes the number of approvals by phase.
Analysis by Therapeutic Area
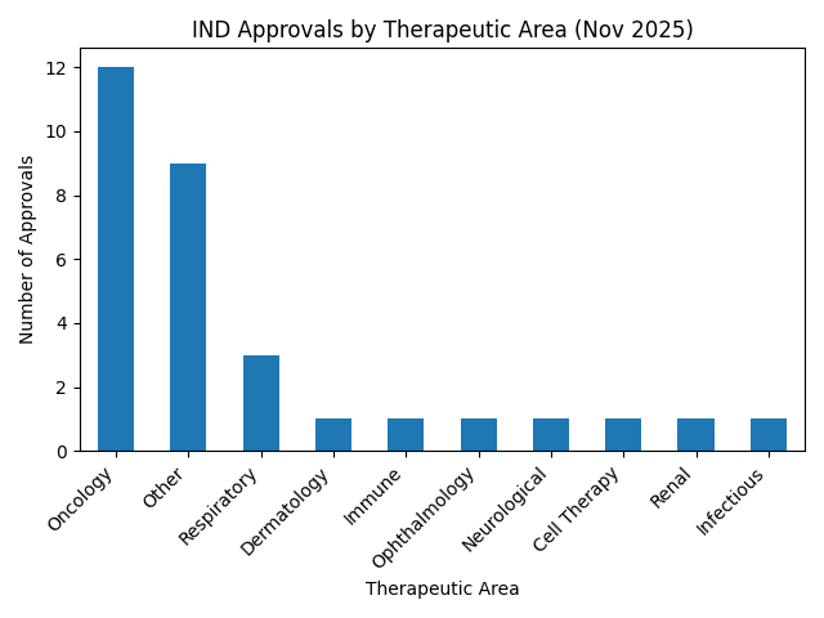
When we examine which therapeutic areas the IND approvals target, cancer leads with 12 approvals among the 31 total. The remaining approvals are distributed across respiratory disease, various rare diseases, dermatology, and a mix of immune, ophthalmology, neurological and cell‑therapy areas. This distribution reaffirms that oncology is at the core of drug development and shows that new immuno‑oncology and targeted therapies are advancing. The chart below summarizes the approvals by therapeutic area.
Status of Domestic Drug Development
Seven of the 10 domestically developed drugs were in Phase 1, demonstrating that Korean companies are expanding their pipelines and focusing on early safety and dosing studies. For example, EHL Bio received approval for a Phase 3 trial of ADSTEM Inj., an autologous adipose‑derived stem‑cell therapy for patients with moderate to severe atopic dermatitis. SK Plasma received approval for a Phase 3 study of an intravenous immunoglobulin (IVIG) formulation for patients with primary immunodeficiency, and the National Cancer Center is conducting a Phase 2 study to evaluate the utility of indocyanine green fluorescent dye in gastric cancer surgery.
Daewon Pharmaceutical and Celltrion each received two IND approvals, the highest among domestic companies. Daewon is running Phase 1 trials on DW4421 and DW5524 for gastroesophageal reflux disease. DW4421 is a potassium‑competitive acid blocker (P‑CAB) designed to inhibit gastric acid secretion, and a confirmatory trial plans to evaluate therapeutic effects in 327 patients; earlier Phase 2 trials showed high healing rates and favorable safety. Celltrion received INDs for CT‑P73, a tissue‑factor‑targeting antibody–drug conjugate (ADC), and CT‑P52, a biosimilar candidate for autoimmune diseases. CT‑P73 links a cytotoxic drug to an antibody to selectively attack cancer cells and aims to be commercialized by 2030.
Other domestic companies also entered early trials. Oscotec is conducting a dose‑escalation Phase 1 study of its in‑house candidate OCT‑598; Addpharma is evaluating the pharmacokinetics and safety of inhalable AD‑230; and Chong Kun Dang is running a Phase 1 study to investigate the pharmacokinetic profile of CKD‑846. The participation of such varied companies shows that Korea’s drug‑development ecosystem is steadily expanding.
Status of Overseas Drug Development
Among the 21 internationally developed drugs, eight are in Phase 3, six in Phase 2, three in Phase 1/2, two in Phase 3b, and one each in Phases 1 and 2a. Janssen and Regeneron obtained INDs for Phase 3 trials targeting ulcerative colitis and multiple myeloma, respectively. GSK received approval for a Phase 3b study of depemokimab, an antibody for severe asthma, and Boehringer Ingelheim began a long‑term trial of BI 1015550 for idiopathic pulmonary fibrosis. These patterns indicate that multinational firms are strategically using Korea for late‑stage clinical validation.
Prominent innovation projects among the overseas approvals include Gilead’s KITE‑363 and BMS Korea’s KarXT. KITE‑363 is an autologous CAR‑T therapy that targets CD19 and CD20 and is being tested in a multicountry Phase 1a/1b trial to assess safety and efficacy. KarXT is a long‑term extension study combining xanomeline with trospium chloride to relieve agitation in patients with Alzheimer’s disease. In addition, Parexel’s Phase 2 study on the leukemia drug revumenib, Revolution Medicines’ Phase 1/2 study of a RAS‑targeted cancer combination, and PPD’s Phase 3 study of an ATTR amyloidosis therapy are among the notable trials taking place in Korea.
Detailed Statistics and Analysis
For domestic drugs, seven approvals were in Phase 1, one in Phase 2 and two in Phase 3. The Phase 1 trials cover a variety of modalities—including ADCs, biosimilars and targeted therapies—highlighting the diversity of early research. Among the 21 foreign approvals there were eight Phase 3, six Phase 2, three Phase 1/2, two Phase 3b, one Phase 1 and one Phase 2a study, underscoring the emphasis on late‑stage research. Many of these Phase 3 trials involve sponsors such as Janssen, Regeneron, PPD, IQVIA, GSK and Boehringer Ingelheim, demonstrating that Korea serves as a hub for large‑scale patient recruitment and efficacy verification.
Key Clinical Examples
EHL Bio’s ADSTEM makes an injectable from a patient’s own adipose tissue stem cells and administers it to people with chronic atopic dermatitis. SK Plasma’s IVIG Phase 3 study confirms the efficacy and safety of an intravenous immunoglobulin formulation for primary immunodeficiency, aiming to localize treatment for rare diseases. The National Cancer Center is analysing the precision of using near‑infrared dye indocyanine green to mark tumour location during laparoscopic gastric‑cancer surgery and its impact on recovery.
International examples include Parexel’s Phase 2 study of revumenib, Revolution Medicines’ Phase 1/2 study of a RAS‑targeted combination, and various trials led by PPD for ATTR amyloidosis and pulmonary hypertension. Infectious‑disease research is also represented, with studies targeting COVID‑19 and other conditions, demonstrating the breadth of therapeutic areas involved.
Technology Trends and Implications
The November approvals highlight cutting‑edge modalities such as antibody therapies, cell therapies and ADCs. An ADC attaches a cytotoxic drug to an antibody to precisely deliver the drug to a target cell; Celltrion’s CT‑P73 is a leading example. CAR‑T cell therapy genetically engineers a patient’s T cells to attack cancer or autoreactive cells, and Gilead’s KITE‑363 exemplifies this approach. While these advanced therapies entail complex manufacturing and high costs, Korea supports their development through strong GMP facilities and clinical experience.
Comparing domestic and foreign IND approvals, 70 % of domestic approvals are in Phase 1, focusing on novel mechanisms and early safety assessment, whereas 67 % of foreign approvals are in Phases 3 and 2. This demonstrates that Korea hosts a unique ecosystem where early and late research coexist. Domestic researchers can gain experience by participating in late‑stage trials of international drugs, and patients benefit from early access to cutting‑edge therapies.
Conclusion and Outlook
The November MFDS IND approval status shows that Korea’s pharmaceutical and biotech industry is building a broad clinical ecosystem that spans early and late development. Domestic companies are expanding their pipelines with Phase 1 trials in oncology, immunology and rare diseases, while global firms leverage Korea for late‑stage studies. These trends position Korea as a global clinical‑trial hub beyond Asia and suggest that collaborations with overseas companies will continue to grow. This analysis can serve as a reference for researchers and companies to understand the Korean clinical‑trial environment and to formulate their strategies, and we plan to continue updating these data in future months.
Intoinworld offers professional CRO services and new‑drug development consulting. If you would like to discuss a specific project, please contact us via the Request a Quote banner, and if you would like to receive the latest industry news and analysis reports, subscribe via the Newsletter Sign‑Up banner. We will continue to provide insights based on MFDS IND approval data.



