Introduction
The MFDS IND approval status 2024 dataset tracks 538 investigational new drug (IND) approvals by the Korean Ministry of Food and Drug Safety (MFDS) between 1 January and 31 December 2024. Understanding this dataset helps sponsors and contract research organizations (CROs) assess the scale and timing of clinical trials in Korea.
Korea introduced the IND system on 3 December 2002, separating clinical trial approval from the new drug application (NDA) process and simplifying dossier requirements. This reform turned Korea into a major destination for clinical research, approving 711 INDs in 2022. The review procedure involves five steps—receipt of application, consultation with the Drug Evaluation Department, dossier review, notification of the result and request for supplementation or final decision—and the statutory review period is 30 working days.
Monthly Approval Trends
MFDS approved 538 INDs in 2024. Monthly approvals peaked in July (55) and September (52) and were lowest in October (34). Approvals remained steady around 44–48 in the first half of the year and 40–44 toward the end.
| Month (YYYY‑MM) | Approvals |
| 2024‑01 | 48 |
| 2024‑02 | 44 |
| 2024‑03 | 45 |
| 2024‑04 | 48 |
| 2024‑05 | 37 |
| 2024‑06 | 44 |
| 2024‑07 | 55 |
| 2024‑08 | 47 |
| 2024‑09 | 52 |
| 2024‑10 | 34 |
| 2024‑11 | 44 |
| 2024‑12 | 40 |
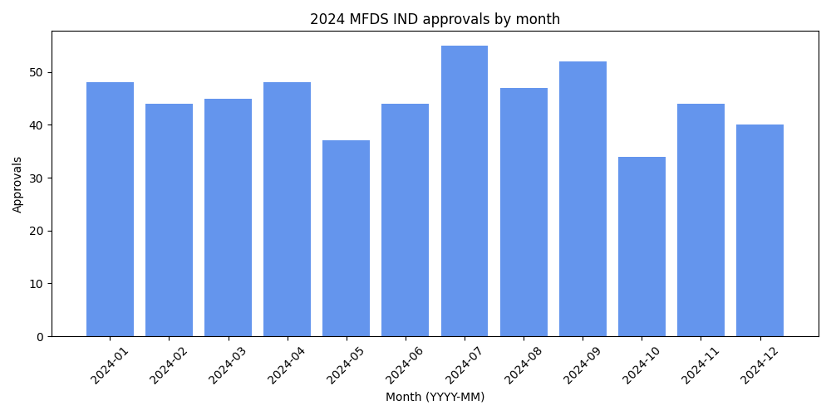
The chart shows that activity surged in mid‑year, suggesting that sponsors concentrated their trial start‑ups around July and September. Early‑ and late‑year approvals were more consistent, implying a stable pipeline of trials throughout the year.
Distribution by Clinical Phase
Phase III approvals were the largest category at 198 approvals (36.8%), followed by Phase I (128 approvals, 23.8%) and Phase II (100 approvals, 18.6%). Early combined phases (Phase 1/2 and 1/2a) accounted for 56 approvals, reflecting increased use of adaptive designs and accelerated early development.
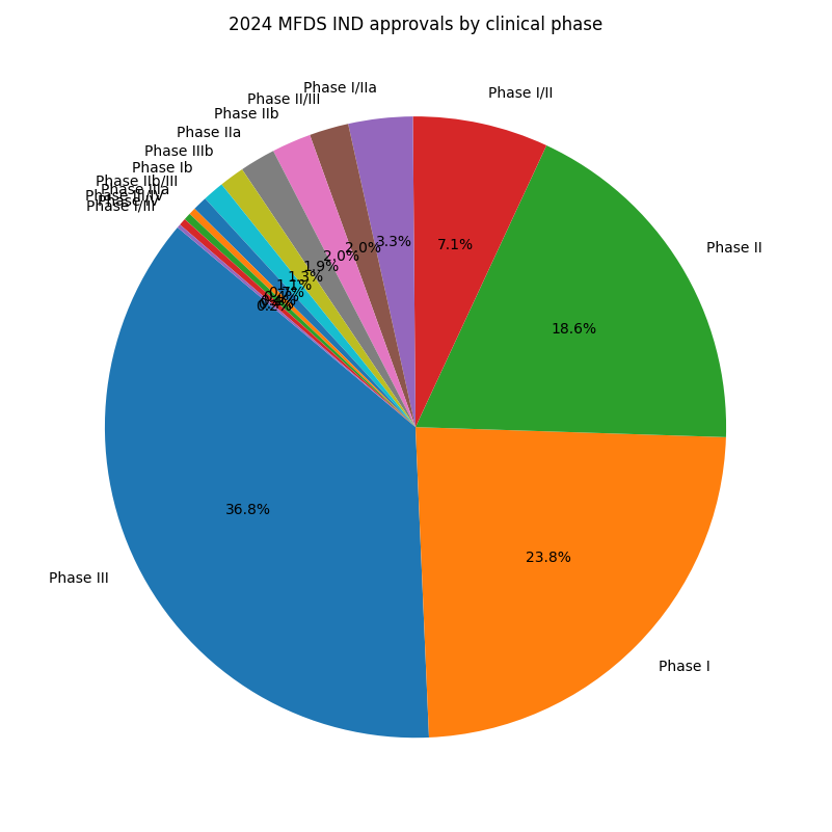
The dominance of Phase III indicates that Korea remains a preferred location for late‑stage trials, but the significant share of Phase I and II trials shows a robust early development environment. Early‑phase combination trials are increasingly common as sponsors seek faster proof‑of‑concept.
Domestic vs International Development and Leading Sponsors
Approvals for internationally developed products numbered 353 (about 65 %), while domestically developed products accounted for 185 approvals (about 35 %). Korea’s clinical‑trial ecosystem thus continues to attract global sponsors, but domestic companies are steadily increasing their share. The top sponsors were IQVIA Korea (39 approvals), MSD Korea (36), Parexel Korea (25), PPD Development Pte Ltd (24) and AstraZeneca Korea (22), showing that major CROs and multinational pharmaceutical firms dominate the approval landscape.
| Category | Approvals |
| Internationally developed trials | 353 |
| Domestically developed trials | 185 |
An IND application in Korea consists of five sequential steps: submission of dossiers, consultation with the Drug Evaluation Department, dossier review, notification of the review result and a request for supplementation or final decision. The review period is 30 working days. Applicants register an account on the MFDS drug information portal (nedrug.mfds.go.kr) and submit the development plan, protocol, manufacture and quality data, non‑clinical and clinical experience data, information on clinical trial institutions, compensation rules and informed consent forms. Trials using already‑approved marketed drugs within approved indications do not require an IND.
Prior to 2002 the IND was part of the NDA process and applicants had to provide full manufacturing and GMP documentation. The introduction of the dedicated IND system simplified the process and has helped Korea become one of the top ten countries for clinical trial volume.
Analysis and Implications
The 2024 MFDS IND approval data shows that Korea remains a key destination for global clinical development. International sponsors continue to dominate the IND landscape, but domestic sponsors are gaining momentum. The high proportion of Phase III approvals signals confidence in Korea’s regulatory framework for late‑stage trials, while the notable number of early‑phase approvals points to vibrant innovation. Leading CROs and multinational pharmaceutical companies drive many approvals, underscoring the importance of experienced partners when navigating Korean regulatory requirements.
This dataset covers approvals up to 31 December 2024; additional approvals in early 2025 will be reported by MFDS later. A detailed Excel file listing all approvals accompanies this article, providing granular information on each trial.
If you plan to initiate a clinical trial in Korea or seek regulatory and operational support, please contact our business development team for a quote request at bd@intoinworld.kr. To stay informed about regulatory updates, clinical‑trial news and market analysis, subscribe to our newsletter by clicking subscribe and submitting your email.



