1. Introduction: The Significance of Korea’s CDx Co‑Development in 2025
Korea’s CDx co‑development has emerged as a key agenda item for the pharmaceutical and biotech industry in 2025. CDx co‑development means developing a new drug and a companion diagnostic device on the same timeline and obtaining approval together. With the implementation of the In‑Vitro Diagnostic Medical Device Act and the establishment of a system for joint review, the companion diagnostic is no longer a peripheral accessory to a drug but an essential partner for therapeutic efficacy and safety.
Recently the MFDS issued additional guidance for CDx conformity assessment and launched an overhaul of the new drug review system in January 2025, shortening the approval period from 420 days to 295 days. These changes are critical considerations when devising a co‑development strategy for domestic and overseas sponsors.
2. AV‑CP‑CL: The Three Pillars of Companion Diagnostic Development
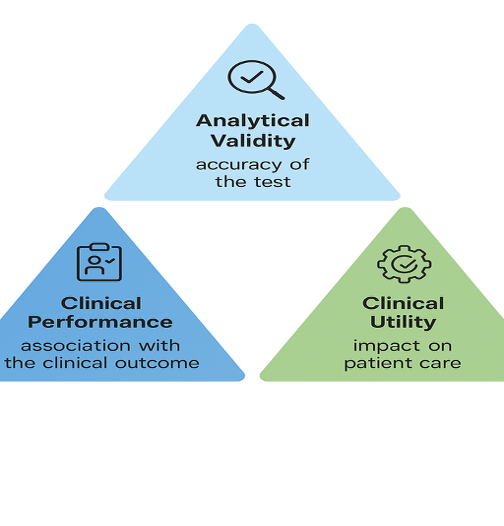
Development of a companion diagnostic consists of three pillars: analytical validity (AV), clinical performance (CP) and clinical utility (CL). Foundation Medicine in the US explains that a CDx must satisfy “analytical validity, clinical validity and clinical utility” to obtain FDA approval. The meaning of each pillar is as follows.
1) Analytical validity (AV): The ability of a test device to accurately and consistently detect the desired biomarker. Indicators such as accuracy, precision, limit of detection (LoD), interference and reproducibility are checked.
2) Clinical performance (CP): Evaluates how well the test results correspond to the actual diagnosis using patient data. Indicators such as sensitivity, specificity and positive/negative agreement are used.
3) Clinical utility (CL): Demonstrates whether test results positively influence treatment decisions and patient outcomes.
3. MFDS Regulations and System Reforms in 2025
3.1 Definition and Classification of CDx
The MFDS defines a companion diagnostic as “a test system that predicts drug responsiveness and safety for a specific patient”, emphasizing its role in predicting therapeutic effects, adverse event risk and determining dosage and administration. In‑vitro diagnostic devices (IVDs) are classified into four risk classes, and CDx fall under class 2 or 3. The review pathway depends on the class, and new products are classified as class 3, requiring joint review of the drug and the device.
3.2 Reform of the New Drug Review System (2025)
In January 2025 the MFDS drastically reformed the new drug approval and review system, reducing the average review period from 420 days to 295 days. Dedicated review teams for each product were established, up to ten face‑to‑face meetings between developers and reviewers were allowed, rolling reviews were introduced so that evaluation begins immediately upon data submission, and clinical and GMP inspections are conducted in parallel. These measures accelerate the timeline and improve predictability for co‑development of drugs and companion diagnostics.
3.3 Strengths of the Korean Clinical Environment
Korea, with Seoul at its center, is one of the most active countries in conducting clinical trials, and even when global clinical trial numbers declined in 2023, Korea’s trials increased by 9%. Nationwide university hospitals and comprehensive cancer centers provide robust infrastructure for multidisciplinary trials. Information technology infrastructure such as electronic medical records also provides advantages in patient recruitment and data management. This environment offers competitiveness in conducting Clinical Performance Tests (CPT) during CDx‑drug co‑development.
4. Checklist for Drug–Diagnostic Co‑Development
Developing an in‑vitro diagnostic device and a drug on the same timeline requires meticulous planning. The following is a reconstructed checklist based on Intoinworld’s report.
1) Pre‑consultation and terminology alignment: Agree on the definitions of patient population, biomarker cut‑off, test methods and platforms, and software version before finalizing the clinical protocol.
2) Label concordance mapping: Prepare a mapping table that aligns the patient population/method on the drug label with the intended use and version of the CDx label on a 1:1 basis.
3) Design of the Clinical Performance Test (CPT): Secure qualified sites to conduct domestic CPTs and design strategies for sample enrichment and multi‑site reproducibility.
4) Assessment of NGS certification: When using NGS environments recognized in Korea, some procedures may be waived, so confirm eligibility during early consultations.
5) Structuring the document package: Compile an analytical variable dictionary, data preprocessing process, statistical analysis plan, audit trails, etc., into a package to enhance reproducibility.
6) Change management plan: Define future change scenarios such as algorithm improvements, platform changes and cut‑off adjustments, and prepare equivalence demonstration plans.
7) Timeline and query management: Manage the drug development and CDx development schedules on a single calendar and prepare a list of anticipated questions to unify responses.
8) Data governance and security: Establish sample chain of custody, version control, access control and log management to ensure traceability and security of data.
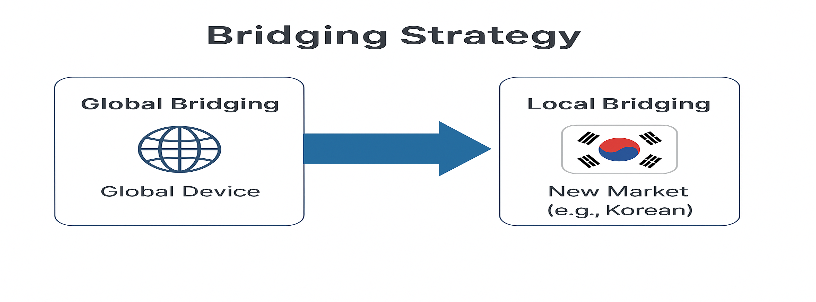
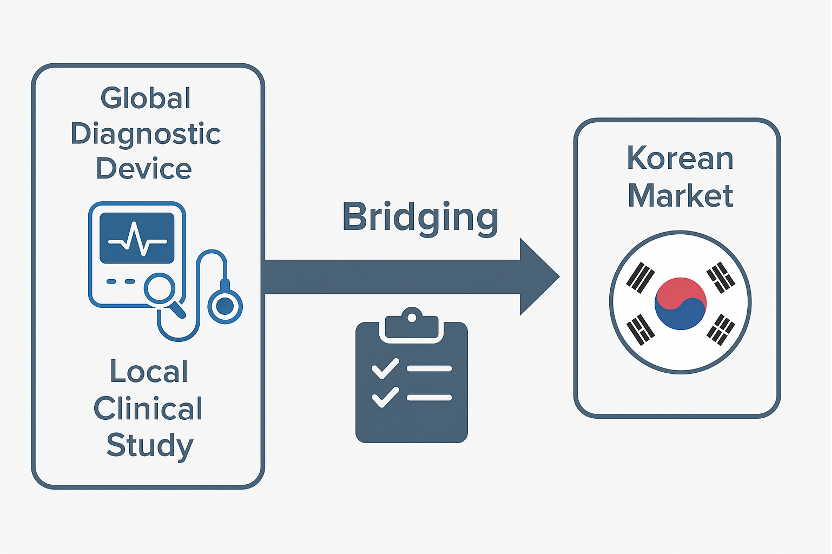
5. Overseas CDx Bridging Strategy
When introducing a CDx already approved overseas into the Korean market, analytical equivalence and clinical linkage consistency must be demonstrated. The accuracy and detection limit proven overseas must be replicated in the Korean setting, and evidence must show that the patient population definitions and cut‑offs targeted by the device apply identically to Korean patients. Any gaps not sufficiently covered by overseas data should be supplemented with domestic CPT data, and terminology and versions on drug and device labels should be harmonized in advance. If a certified NGS platform is used, certain requirements may be waived, but the certification scope must be clearly identified and supporting documents prepared.
The diagram below summarizes the bridging process for introducing an overseas‑approved device into the Korean environment.
6. Strategic Value of Korean CROs and CDMOs
In the development of drugs and CDx, the roles of CROs (contract research organizations) and CDMOs (contract development and manufacturing organizations) are essential. The CRO industry has about a 40‑year history in Korea and is an important pillar: roughly 70% of the domestic CRO market is occupied by global CROs and 30% by approximately 20–30 domestic CROs including Intoinworld. Domestic CROs provide services optimized to Korean regulations and hospital networks, helping overseas sponsors meet local regulatory requirements and overcome language and cultural barriers. CDMOs such as Samsung Biologics and Lotte Biologics, world‑class companies, support production of clinical‑grade prototypes and preparation for commercialization, enabling global pharmaceutical companies to carry out clinical development and production simultaneously in Korea.
Pharmaceutical and biotech companies planning co‑development projects in Korea can work with domestic CROs like Intoinworld to devise a fast and efficient regulatory and clinical execution strategy. For more details, see Intoinworld’s clinical trial information page.
7. Conclusion and Next Steps
Co‑development of companion diagnostics and new drugs has become essential for providing optimal treatment to patients. With the MFDS reform and growth of Korea’s clinical environment in 2025, the regulatory requirements and procedures needed for co‑development have become clearer and the timeline shorter. If evidence is prepared based on the AV‑CP‑CL concept and the checklist is used to ensure document consistency, CPT design and change management planning, it is possible to conduct joint reviews efficiently in Korea. When introducing overseas CDx into the Korean market, a bridging strategy must be prepared to demonstrate analytical performance and clinical linkage. To navigate these complex processes successfully, partners with expertise in local regulations and clinical environments are indispensable.
Work with Intoinworld
Request a quote: If you are preparing for a new CDx development or clinical project, request a free quote to consult with Intoinworld experts.
Subscribe to the newsletter: To stay on top of the latest clinical trial trends and regulatory changes, subscribe to Intoinworld’s newsletter for regular updates.
FAQ
Q1. What is a companion diagnostic (CDx) and why is it important?
A1. A companion diagnostic is a device that tests a patient’s biomarker to predict the safety and efficacy of a specific drug. The FDA emphasizes that CDx must possess analytical validity (accurate biomarker detection), clinical validity (predicting patient response) and clinical utility (informing treatment decisions). Without CDx, suitable patients may not be identified, reducing the therapeutic benefits of a new drug.
Q2. What is the most important point to consider when carrying out co‑development in Korea?
A2. To successfully conduct co‑development domestically, developers must understand the MFDS classification and regulations and, from early stages, have the drug development and diagnostic development teams jointly discuss patient population definitions and testing methods. The checklist, including label concordance mapping, CPT design, NGS certification and change management plans, should be referenced to structure the document package. The new MFDS review system shortens the review period but requires completeness and consistency of submitted documents.
Q3. What data are required when bringing an overseas‑approved CDx into Korea?
A3. To have an overseas‑approved device recognized in Korea, analytical performance equivalence and clinical linkage consistency must be demonstrated. That is, it must be shown that the accuracy, precision and detection limits proven overseas are reproduced in Korean patients and testing environments, and that the patient population definitions and cut‑off values targeted by the device apply identically in Korean clinical settings. Any portions not sufficiently supported by overseas data should be supplemented with domestic CPTs, and terminology and versions on the drug and device labels should be harmonized in advance.



