Korea’s drug classification rule was updated in February 2026. But this was not a major system overhaul.
On February 11, 2026, South Korea’s Ministry of Food and Drug Safety (MFDS) issued an administrative notice for a partial amendment to the Rule on Classification Numbers for Pharmaceuticals. The amendment was then formally adopted on February 23, 2026, as MFDS Rule No. 223. The core reason was straightforward: the rule’s validity period was approaching its end, and MFDS decided that the rule should continue to remain in force.
This was not a headline-grabbing reform of Korea’s pharmaceutical classification framework. It was a continuity measure. In practical terms, MFDS was not changing the logic of the classification system. It was confirming that the existing framework still needed to be maintained.
For companies outside Korea, this kind of update can look minor. But in regulatory work, small administrative rules often function like load-bearing walls in a building. They do not always attract attention, yet they support the consistency of product classification, regulatory communication, and internal data mapping.
What is the Rule on Classification Numbers for Pharmaceuticals?
The Rule on Classification Numbers for Pharmaceuticals is an MFDS administrative rule used in Korea’s pharmaceutical management system. At a basic level, it provides the classification-number structure used for pharmaceuticals and related regulated products. The rule itself has existed for many years within Korea’s administrative rule system, which is why the 2026 amendment was framed not as a redesign, but as an extension of continued applicability.
For overseas readers, the easiest way to understand this is to think of classification numbers as part of the regulatory language of the market. They are not just labels. They help support how products are organized, referenced, and consistently understood across administrative and regulatory processes.
That is also why even a limited amendment can still be relevant. A market-entry team may not need this rule every day, but when a company is aligning internal product databases, building submission logic, or checking how products are categorized in the Korean system, framework stability matters.
What exactly changed in February 2026?
The February 2026 amendment was narrow in scope, but it is still worth explaining precisely.
MFDS first issued an administrative notice on February 11, 2026, stating that the purpose of the amendment was to extend the validity period of the Rule on Classification Numbers for Pharmaceuticals because the existing validity period was nearing its end and the rule was still considered necessary under current conditions. The amendment was then formally finalized on February 23, 2026 as MFDS Rule No. 223. In the official amendment summary, MFDS identified the main change as an extension of the rule’s validity period within the three-year range allowed under the Korean rule governing the issuance and management of directives and administrative rules, and referred specifically to Article 2 and related provisions.
That means the practical update was not a redesign of the classification-number framework itself. MFDS did not announce a new classification logic, a restructuring of pharmaceutical categories, or a new scope of regulated products in this amendment. Instead, the agency kept the existing framework in place by extending the rule’s legal-operational life.
The easiest way to see the change is through a before-and-after comparison.
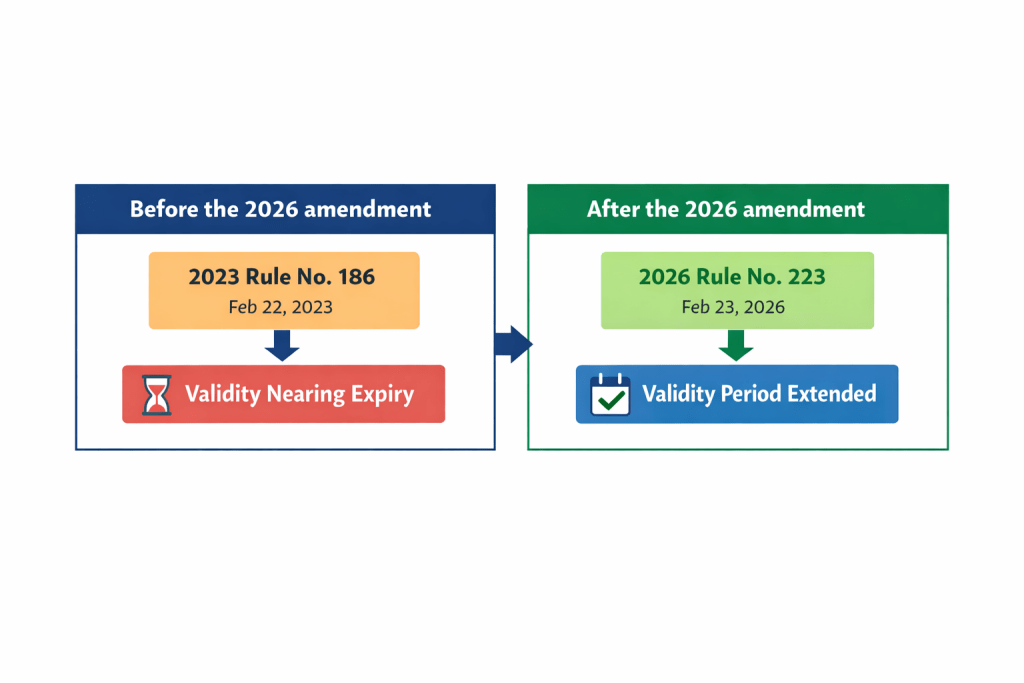
Before the 2026 amendment: the rule was operating under the earlier version, including the 2023 partial amendment (MFDS Rule No. 186, dated February 22, 2023), and its validity period was approaching expiry. MFDS had already used the same legal mechanism in 2023 to extend the rule’s validity when it concluded that the framework still needed to remain in force.
After the 2026 amendment: the underlying classification framework remained in place, but its validity period was extended again so that the rule could continue to operate without interruption. In other words, the structure stayed the same, while the period of continued applicability was refreshed.
From a regulatory-intelligence perspective, that distinction matters. A company reading only the title of the amendment might assume that MFDS had revised how pharmaceutical classification numbers are assigned or interpreted. But the official materials point in a different direction: the regulator chose continuity over redesign.
Why does a small amendment still matter?
The first reason is regulatory predictability.
When a regulator extends the validity of an existing administrative rule rather than replacing it, the immediate message is stability. For pharmaceutical companies, especially those entering Korea from abroad, stable administrative architecture reduces one layer of uncertainty. It suggests that companies do not need to reframe their understanding of the underlying classification structure in the near term. This is an inference based on the amendment’s continuity-focused rationale rather than a direct MFDS statement, but it is a practical one.
The second reason is submission and internal alignment.
Regulatory work is not only about major approval pathways. It is also about internal consistency. A classification rule may affect how teams map products, describe categories, organize master data, or ensure that internal terminology matches the language used in the Korean regulatory environment. The smaller the rule appears, the easier it is for global teams to ignore it. That is often a mistake.
The third reason is market literacy.
Many overseas sponsors understand headline Korean drug policies such as review acceleration, digitalization, or rare-disease initiatives. But they are less familiar with the lower-level administrative rules that shape day-to-day regulatory practice. This amendment is a useful reminder that Korea’s regulatory environment is built not only on major reforms, but also on the steady maintenance of background rules that keep the system coherent.
Conclusion
The February 2026 amendment to Korea’s Rule on Classification Numbers for Pharmaceuticals was modest in scope, but still meaningful in context. MFDS announced the amendment on February 11, 2026, and finalized it on February 23, 2026, primarily to extend the rule’s validity period. That may sound administrative, but the signal behind it is clear: the existing classification-number framework continues to matter within Korea’s pharmaceutical regulatory system.
If your team is exploring clinical trials in Korea, regulatory planning, or Korea market-entry strategy, Intoinworld can help you navigate the local landscape with practical, business-focused insight. You are welcome to submit a quotation request anytime to learn more about how we support Korea-based clinical development and regulatory execution.
If you would also like to stay updated on Korea’s regulatory and industry trends, subscribe to the Intoinworld newsletter to receive the latest insights, policy developments, and market signals.



